|
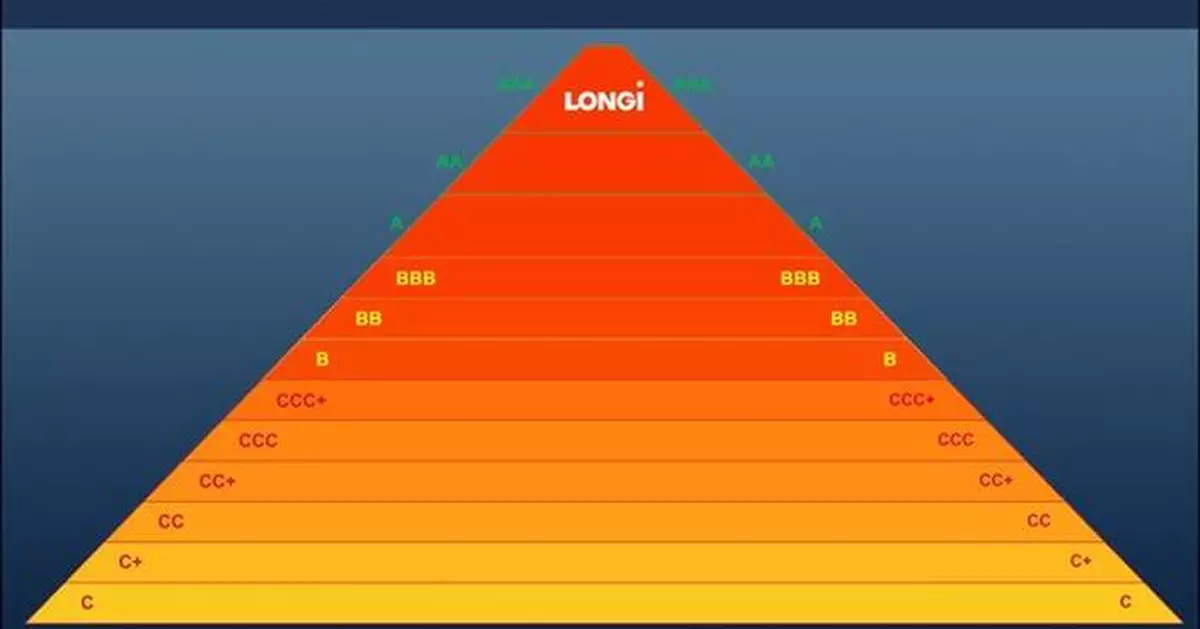
XI'AN, China, April 17, 2025 /PRNewswire/ -- LONGi has announced the retention of its AAA rating in PV-Tech's Q1 2025 PV ModuleTech bankability ratings, marking the company's 21st consecutive quarter at this top tier and underscoring its long-term commitment to quality, innovation, financial stability and leadership in the photovoltaic industry.
The PV ModuleTech bankability ratings are derived from a thorough evaluation process that examines manufacturers across several critical dimensions, including value chain strength, production capacity, global shipment profiles, capital expenditure (capex), R&D investment and cash-flow management. LONGi's consistent AAA status reflects not only its robust operational metrics but also its dedication to advancing solar technology.
Relying on its profound technology accumulation and advanced intelligent manufacturing advantages, LONGi has made continuous breakthroughs in HPBC technology and forward-looking R&D, and its products for utility and distributed scenarios have been implemented successively.
Since the launch of the new-generation BC products based on HPBC 2.0 in May 2024, LONGi has been winning large orders globally.
At the beginning of April, LONGi signed a 226MW Hi-MO 9 module supply agreement in Greece, which marks the largest BC utility project in Europe.
At the end of March, LONGi signed a Hi-MO X10 module supply framework agreement with a total capacity of 555MW with multiple EPC companies at the South Africa International Photovoltaic and Energy Storage Exhibition. The high-power generation per unit area and anti-shading features of the products have been highly recognized by African customers.
By the end of the first quarter of 2025, the global shipments of BC modules had exceeded 30GW, and the scale of reserved orders had exceeded 40GW.
With technological innovation as its mission, LONGi has been constantly improving the R&D of conversion efficiency in the crystalline silicon and crystalline silicon-perovskite tandem tracks. Up to now, it still holds the world records in these two major tracks.
On April 11th, LONGi announced at its Wuhu base in Anhui Province, China: Through the authoritative certification of the Institute for Solar Energy Research Hamelin (ISFH) in Germany, the photoelectric conversion efficiency of its independently developed Hybrid Interdigitated-Back-Contact (HIBC) crystalline silicon solar cell has reached 27.81%, pushing the exploration of the limits of monocrystalline silicon photovoltaic cells to a new height.
At the same time, LONGi independently developed a two-terminal crystalline silicon-perovskite tandem solar cell that has achieved a conversion efficiency of 34.85%, certified by the U.S. National Renewable Energy Laboratory (NREL), once again breaking the world record for crystalline silicon-perovskite tandem cell efficiency.
The 21 consecutive AAA ratings are not only a strong testament to LONGi's reliability but also a footnote to LONGi's consistent commitment to product quality, stable operation, and long-termism. In the future, LONGi will continue to enhance its technological strength and risk-resistance capabilities, and work hand in hand with global partners to create a better future.
XI'AN, China, April 17, 2025 /PRNewswire/ -- LONGi has announced the retention of its AAA rating in PV-Tech's Q1 2025 PV ModuleTech bankability ratings, marking the company's 21st consecutive quarter at this top tier and underscoring its long-term commitment to quality, innovation, financial stability and leadership in the photovoltaic industry.
The PV ModuleTech bankability ratings are derived from a thorough evaluation process that examines manufacturers across several critical dimensions, including value chain strength, production capacity, global shipment profiles, capital expenditure (capex), R&D investment and cash-flow management. LONGi's consistent AAA status reflects not only its robust operational metrics but also its dedication to advancing solar technology.
Relying on its profound technology accumulation and advanced intelligent manufacturing advantages, LONGi has made continuous breakthroughs in HPBC technology and forward-looking R&D, and its products for utility and distributed scenarios have been implemented successively.
Since the launch of the new-generation BC products based on HPBC 2.0 in May 2024, LONGi has been winning large orders globally.
At the beginning of April, LONGi signed a 226MW Hi-MO 9 module supply agreement in Greece, which marks the largest BC utility project in Europe.
At the end of March, LONGi signed a Hi-MO X10 module supply framework agreement with a total capacity of 555MW with multiple EPC companies at the South Africa International Photovoltaic and Energy Storage Exhibition. The high-power generation per unit area and anti-shading features of the products have been highly recognized by African customers.
By the end of the first quarter of 2025, the global shipments of BC modules had exceeded 30GW, and the scale of reserved orders had exceeded 40GW.
With technological innovation as its mission, LONGi has been constantly improving the R&D of conversion efficiency in the crystalline silicon and crystalline silicon-perovskite tandem tracks. Up to now, it still holds the world records in these two major tracks.
On April 11th, LONGi announced at its Wuhu base in Anhui Province, China: Through the authoritative certification of the Institute for Solar Energy Research Hamelin (ISFH) in Germany, the photoelectric conversion efficiency of its independently developed Hybrid Interdigitated-Back-Contact (HIBC) crystalline silicon solar cell has reached 27.81%, pushing the exploration of the limits of monocrystalline silicon photovoltaic cells to a new height.
At the same time, LONGi independently developed a two-terminal crystalline silicon-perovskite tandem solar cell that has achieved a conversion efficiency of 34.85%, certified by the U.S. National Renewable Energy Laboratory (NREL), once again breaking the world record for crystalline silicon-perovskite tandem cell efficiency.
The 21 consecutive AAA ratings are not only a strong testament to LONGi's reliability but also a footnote to LONGi's consistent commitment to product quality, stable operation, and long-termism. In the future, LONGi will continue to enhance its technological strength and risk-resistance capabilities, and work hand in hand with global partners to create a better future.
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **
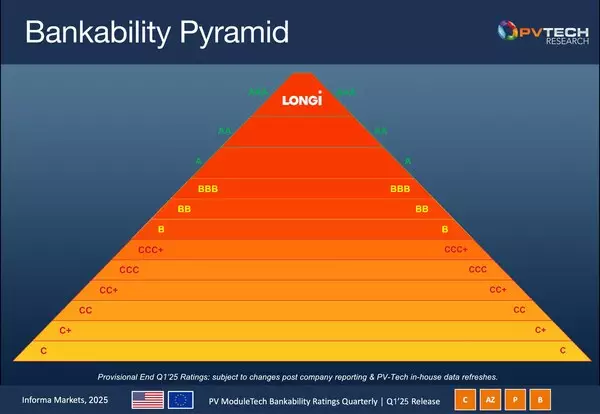
LONGi maintains AAA ranking in Q1 2025 PV ModuleTech Bankability Ratings
|
ROCKVILLE, Md., May 8, 2026 /PRNewswire/ -- USA -- AAVnerGene Inc. today announced the launch of AAVone®2.1, the next-generation evolution of its proprietary AAVone® single-plasmid AAV production system, designed to improve AAV vector productivity, full-capsid yield, manufacturing efficiency, and cost effectiveness.
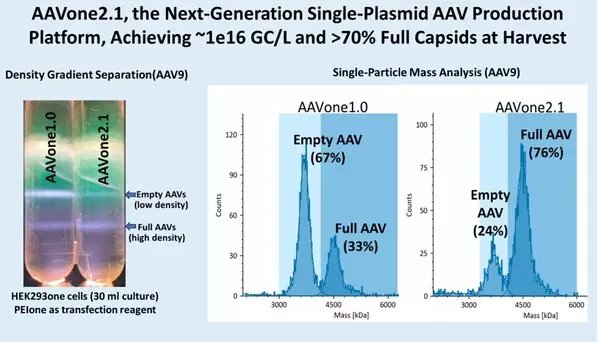
AAVone®2.1 has achieved ~1x1016 genome copies (GC)/L of cell culture and greater than 70% full capsids at harvest, representing a significant advance over AAVone®1.0 and conventional multi-plasmid AAV production systems, which often generate high proportions of empty capsids. The platform has demonstrated performance across multiple AAV serotypes and compatibility with HEK293-based cell lines and existing manufacturing workflows.
These improvements may reduce plasmid requirements, culture volume, purification burden, processing steps, processing time, and manufacturing cost, while improving overall vector quality. By addressing key constraints in AAV production, AAVone®2.1 is designed to support the clinical and commercial scalability of AAV-based gene therapy programs.
"AAV manufacturing remains one of the most important technical and economic challenges in gene therapy," said Qizhao Wang, Ph.D., Chief Technology Officer of AAVnerGene. "Conventional multi-plasmid systems have enabled the field for decades, but limitations in productivity, empty capsid burden, and purification complexity continue to create barriers to development and commercialization. AAVone®2.1 was developed to address these challenges through a simplified single-plasmid system that improves vector productivity and full-capsid ratio. "
"AAV gene therapy continues to face commercialization challenges, with manufacturing cost, vector quality, and process scalability among the most critical constraints," said Daozhan Yu, PhD, Chief Executive Officer of AAVnerGene. "AAVone®2.1 has the potential to make AAV manufacturing simpler, more scalable, and more cost-efficient, helping more AAV gene therapy programs become clinically and commercially viable."
AAVnerGene's partners have licensed the technology and are developing multiple AAV gene therapy programs using the platform. The company's goal is to provide enabling technologies that help overcome manufacturing barriers, reduce development costs, and expand the number of gene therapy programs that can reach patients.
About AAVnerGene Inc.
AAVnerGene is a Maryland-based biotech company developing solutions to address bottlenecks in AAV gene therapy, including manufacturing, toxicity, qualification, and delivery. Its platforms include AAVone®, AAV-Q (a potency and rcAAV assay platform), and ATHENA (a capsid engineering platform), designed to improve productivity, quality, scalability, efficiency, and safety in AAV gene therapy drug development.
Media & Business Inquiries:
customer@aavnergene.com
www.aavnergene.com
ROCKVILLE, Md., May 8, 2026 /PRNewswire/ -- USA -- AAVnerGene Inc. today announced the launch of AAVone®2.1, the next-generation evolution of its proprietary AAVone® single-plasmid AAV production system, designed to improve AAV vector productivity, full-capsid yield, manufacturing efficiency, and cost effectiveness.
AAVone®2.1 has achieved ~1x1016 genome copies (GC)/L of cell culture and greater than 70% full capsids at harvest, representing a significant advance over AAVone®1.0 and conventional multi-plasmid AAV production systems, which often generate high proportions of empty capsids. The platform has demonstrated performance across multiple AAV serotypes and compatibility with HEK293-based cell lines and existing manufacturing workflows.
These improvements may reduce plasmid requirements, culture volume, purification burden, processing steps, processing time, and manufacturing cost, while improving overall vector quality. By addressing key constraints in AAV production, AAVone®2.1 is designed to support the clinical and commercial scalability of AAV-based gene therapy programs.
"AAV manufacturing remains one of the most important technical and economic challenges in gene therapy," said Qizhao Wang, Ph.D., Chief Technology Officer of AAVnerGene. "Conventional multi-plasmid systems have enabled the field for decades, but limitations in productivity, empty capsid burden, and purification complexity continue to create barriers to development and commercialization. AAVone®2.1 was developed to address these challenges through a simplified single-plasmid system that improves vector productivity and full-capsid ratio. "
"AAV gene therapy continues to face commercialization challenges, with manufacturing cost, vector quality, and process scalability among the most critical constraints," said Daozhan Yu, PhD, Chief Executive Officer of AAVnerGene. "AAVone®2.1 has the potential to make AAV manufacturing simpler, more scalable, and more cost-efficient, helping more AAV gene therapy programs become clinically and commercially viable."
AAVnerGene's partners have licensed the technology and are developing multiple AAV gene therapy programs using the platform. The company's goal is to provide enabling technologies that help overcome manufacturing barriers, reduce development costs, and expand the number of gene therapy programs that can reach patients.
About AAVnerGene Inc.
AAVnerGene is a Maryland-based biotech company developing solutions to address bottlenecks in AAV gene therapy, including manufacturing, toxicity, qualification, and delivery. Its platforms include AAVone®, AAV-Q (a potency and rcAAV assay platform), and ATHENA (a capsid engineering platform), designed to improve productivity, quality, scalability, efficiency, and safety in AAV gene therapy drug development.
Media & Business Inquiries:
customer@aavnergene.com
www.aavnergene.com
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
AAVnerGene Announces AAVone®2.1, a Next-Generation Single-Plasmid AAV Production Platform Achieving ~1e16 GC/L and >70% Full Capsids at Harvest