|
- The bone graft incorporating rhBMP-2 has received FDA IDE approval for a pivotal clinical trial in spinal fusion
- Positioned for global expansion in $750M spinal bone graft market
SEOUL, South Korea, April 24, 2025 /PRNewswire/ -- CGBIO(CEO Hyun Seung Yu), a leading Korean company specializing in bio-regenerative medicine, proudly announces that its innovative bone graft substitute, NOVOSIS PUTTY, has received Investigational Device Exemption (IDE) approval from the U.S. Food and Drug Administration (FDA). This pivotal approval paves the way for a clinical trial in spinal fusion procedures within the United States.
NOVOSIS PUTTY, previously designated as a Breakthrough Device by the FDA in December 2023, leverages novel technology to address unmet needs in bone regeneration. This milestone marks NOVOSIS PUTTY as the first Korean-developed bio-combined medical device to reach this stage in the U.S., signifying a significant step toward Premarket Approval (PMA) and subsequent commercialization.
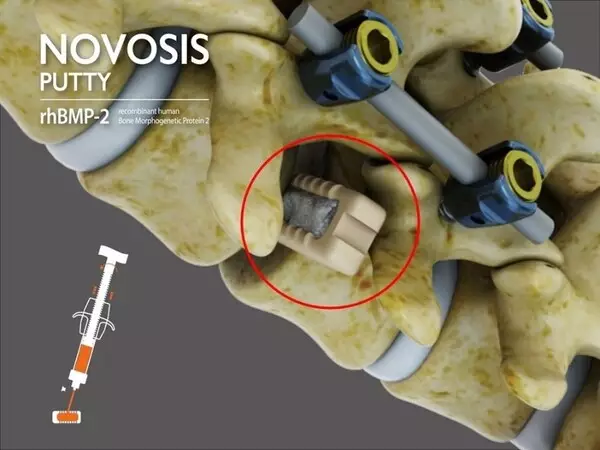
The product features a dual-carrier system utilizing Hydroxyapatite (HA) and Tri-Calcium Phosphate (TCP), combined with CGBIO's proprietary sustained-release technology, SLORELâ„¢, to control the release of recombinant human bone morphogenetic protein-2 (rhBMP-2). This system is engineered to enhance high-density bone formation while minimizing ectopic bone growth, a common adverse effect in earlier rhBMP-2-based products. The safety and efficacy of NOVOSIS PUTTY have been validated in peer-reviewed publications, including the Journal of Clinical Medicine.
The rhBMP-2 protein used in NOVOSIS PUTTY is manufactured by Daewoong Pharmaceutical, a strategic partner of CGBIO. During the IDE application process, the FDA conducted an in-depth review of the manufacturing process and Chemistry, Manufacturing, and Controls (CMC) data, reflecting the FDA's heightened scrutiny for bio-combined implants.
Jumi Han, Head of U.S. Development at CGBIO USA, overseeing the pivotal clinical program, commented, "Securing IDE approval for this pivotal trial validates our global clinical infrastructure and product quality. We are fully committed to executing a robust clinical trial and expanding NOVOSIS PUTTY's reach across the U.S. and other major markets".
Hyun Seung Yu, CEO of CGBIO, stated, "This IDE approval reflects years of consistent R&D efforts and strengthens our global competitiveness. The U.S. bone graft market is notoriously difficult to penetrate, and this achievement underscores the global potential of NOVOSIS PUTTY. We will continue to provide transformative treatment options that improve quality of life for patients worldwide".
In February 2025, CGBIO and its subsidiary CG MedTech signed a partnership agreement with Johnson & Johnson MedTech for the exclusive supply of NOVOSIS and NOVOSIS TRAUMA products across Korea and other Asian territories. The new IDE approval for NOVOSIS PUTTY is expected to accelerate the global expansion of the entire NOVOSIS product family.
About CGBIO
CGBIO is a global medical device company specializing in advanced biomaterials and regenerative medicine technologies. With a robust product portfolio and numerous successful clinical cases, CGBIO is committed to delivering innovative medical solutions that enhance patient outcomes and quality of life. And to support growing gloval demand, CGBio has begun construction of a new manufacturing facility. While Daewoong Bio has already completed a lare-scale plant dedicated to rhBMP-2 production, ensuring stable supply for the upcomming U.S. and global rollout.
- The bone graft incorporating rhBMP-2 has received FDA IDE approval for a pivotal clinical trial in spinal fusion
- Positioned for global expansion in $750M spinal bone graft market
SEOUL, South Korea, April 24, 2025 /PRNewswire/ -- CGBIO(CEO Hyun Seung Yu), a leading Korean company specializing in bio-regenerative medicine, proudly announces that its innovative bone graft substitute, NOVOSIS PUTTY, has received Investigational Device Exemption (IDE) approval from the U.S. Food and Drug Administration (FDA). This pivotal approval paves the way for a clinical trial in spinal fusion procedures within the United States.
NOVOSIS PUTTY, previously designated as a Breakthrough Device by the FDA in December 2023, leverages novel technology to address unmet needs in bone regeneration. This milestone marks NOVOSIS PUTTY as the first Korean-developed bio-combined medical device to reach this stage in the U.S., signifying a significant step toward Premarket Approval (PMA) and subsequent commercialization.
The product features a dual-carrier system utilizing Hydroxyapatite (HA) and Tri-Calcium Phosphate (TCP), combined with CGBIO's proprietary sustained-release technology, SLORELâ„¢, to control the release of recombinant human bone morphogenetic protein-2 (rhBMP-2). This system is engineered to enhance high-density bone formation while minimizing ectopic bone growth, a common adverse effect in earlier rhBMP-2-based products. The safety and efficacy of NOVOSIS PUTTY have been validated in peer-reviewed publications, including the Journal of Clinical Medicine.
The rhBMP-2 protein used in NOVOSIS PUTTY is manufactured by Daewoong Pharmaceutical, a strategic partner of CGBIO. During the IDE application process, the FDA conducted an in-depth review of the manufacturing process and Chemistry, Manufacturing, and Controls (CMC) data, reflecting the FDA's heightened scrutiny for bio-combined implants.
Jumi Han, Head of U.S. Development at CGBIO USA, overseeing the pivotal clinical program, commented, "Securing IDE approval for this pivotal trial validates our global clinical infrastructure and product quality. We are fully committed to executing a robust clinical trial and expanding NOVOSIS PUTTY's reach across the U.S. and other major markets".
Hyun Seung Yu, CEO of CGBIO, stated, "This IDE approval reflects years of consistent R&D efforts and strengthens our global competitiveness. The U.S. bone graft market is notoriously difficult to penetrate, and this achievement underscores the global potential of NOVOSIS PUTTY. We will continue to provide transformative treatment options that improve quality of life for patients worldwide".
In February 2025, CGBIO and its subsidiary CG MedTech signed a partnership agreement with Johnson & Johnson MedTech for the exclusive supply of NOVOSIS and NOVOSIS TRAUMA products across Korea and other Asian territories. The new IDE approval for NOVOSIS PUTTY is expected to accelerate the global expansion of the entire NOVOSIS product family.
About CGBIO
CGBIO is a global medical device company specializing in advanced biomaterials and regenerative medicine technologies. With a robust product portfolio and numerous successful clinical cases, CGBIO is committed to delivering innovative medical solutions that enhance patient outcomes and quality of life. And to support growing gloval demand, CGBio has begun construction of a new manufacturing facility. While Daewoong Bio has already completed a lare-scale plant dedicated to rhBMP-2 production, ensuring stable supply for the upcomming U.S. and global rollout.
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **
CGBIO Receives FDA IDE Approval for NOVOSIS PUTTY, Advancing Toward U.S. Market Entry
Unlocking expressive freedom for serious digital artists with Wacom's latest rotation-sensitive pen—bringing the feel of traditional tools to digital creation.
TOKYO, May 13, 2026 /PRNewswire/ -- Wacom today announced the Wacom Art Pen 2, a rotation-sensitive pen designed for digital artists and creative professionals seeking precise, expressive control in their digital workflows.
Expressive Control with Barrel Rotation
Building on the legacy of the original Wacom Art Pen (KP701E), the new Wacom Art Pen 2 continues to offer 360° barrel rotation and brings this capability into Wacom's latest pen technology. With compatible brushes, pens, and cursors, it can respond not only to pressure and tilt but also to the pen's rotational angle (when used with supported software). The Wacom Art Pen 2 enables artists to vary the thickness and character of their lines simply by rotating the pen in their hand—much like how traditional artists adjust the angle of a pencil, flat brush, felt marker, or calligraphy pen to achieve different expressive effects. This intuitive control allows for a more natural and dynamic drawing experience that closely mirrors the versatility of familiar traditional tools.
"For years, the Wacom Art Pen has been cherished by artists for its unique feel and expressive possibilities. With Wacom Art Pen 2, we've listened to our passionate community and refined what they love, bringing that familiar experience into our latest pen technology," said Koji Yano, Senior Vice President, Creative Experience Unit at Wacom. "By integrating these capabilities into our current devices, we aim to give creators even more freedom to bring their vision to life."
Smart Design for Artists
The Wacom Art Pen 2 features Wacom's battery-free EMR technology and is equipped with three pen buttons, helping streamline workflow and minimize interruptions. With up to 8,192 levels of pressure sensitivity, the pen delivers smooth, precise control from delicate sketches to bold, dynamic strokes. A built-in nib holder inside the pen barrel stores three spare nibs, so artists can quickly replace tips whenever needed.
New Nib Options for Artistic Feel
Artists can choose from three newly developed nib types—Art Pen 2 Carbon Shaft POM Nibs, Art Pen 2 POM Nibs, and Art Pen 2 Felt Nibs—each engineered to deliver a distinct drawing feel. The pen ships with an Art Pen 2 Carbon Shaft POM nib installed, and these nibs are exclusive to Wacom Art Pen 2 and are not interchangeable with Wacom Pro Pen 3 nibs.
Compatibility
Wacom Art Pen 2 is compatible with Wacom MovinkPad Pro 14, Wacom Intuos Pro (PTK470 / PTK670 / PTK870), Wacom Cintiq 16 (DTK168), Wacom Cintiq 24 (DTK246), and Wacom Cintiq 24 touch (DTH246). Wacom Cintiq Pro (DTH172 / DTH227 / DTH271) will be supported later this year.
Availability
The Wacom Art Pen 2 will be available soon.
Note: Unlike the Wacom Pro Pen 3, the Wacom Art Pen 2 does not support interchangeable button plates, grips or balance weights for customizing the pen's grip or center of gravity.
About Wacom
Wacom is a global leader in digital pen solutions, offering intuitive tools for writing and drawing. Our products are trusted across creative fields — from studios and classrooms to healthcare — supporting students and professionals alike. Wacom pen technologies also power many pen-enabled PCs, tablets, and phones through partnerships with top tech brands.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

Wacom Unveils Wacom Art Pen 2: Next-Generation Pen with 360° Barrel Rotation for Digital Artists