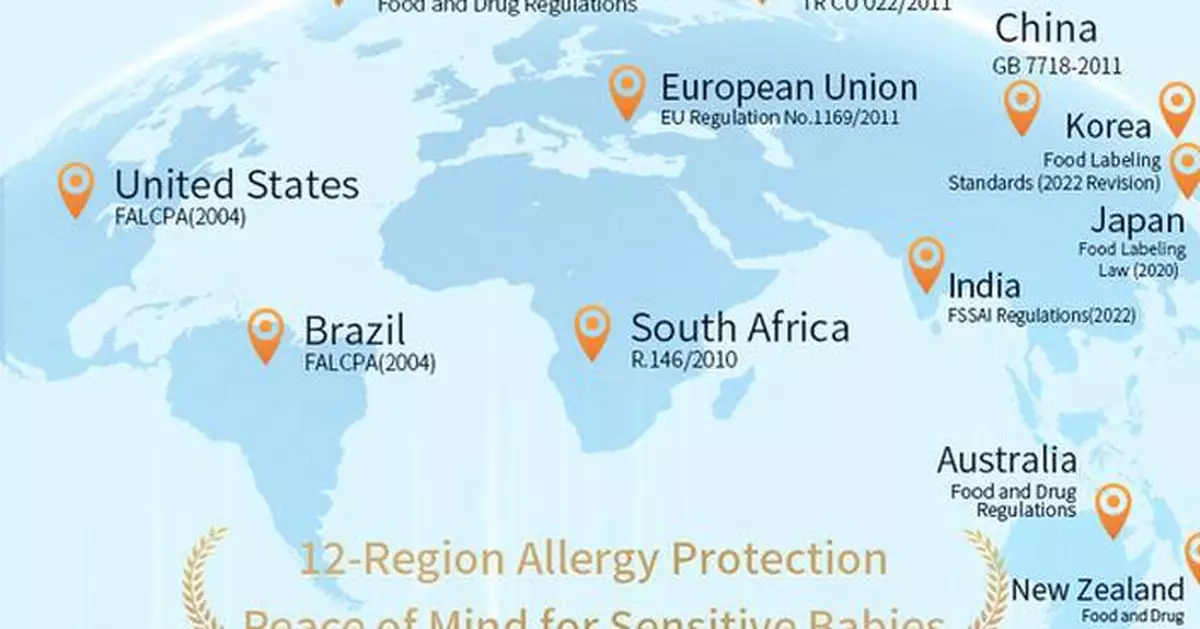
SYDNEY, April 25, 2025 /PRNewswire/ -- Allergy prevention is a concern of parents when they are choosing foods and supplements for their babies. Now, there is a new breakthrough in this dilemma. Recently, Witsbb, an Australian infant nutrition brand, has for the first time released the "Allergen-Free 100+ Transparent List", which can help select nutritional products for sensitive babies. With innovative technology, 100+ common food allergens are eliminated, including but not limited to eight common allergens such as seafood/shrimp shells, milk, eggs, and so on, as pointed out in the National Standard for Food Safety: General Principles for the Labeling of Prepackaged Foods (GB 7718-2011). The list covers common allergens that are mandatorily labeled in 12 countries and regions, such as China, Australia, and the United States, allowing cross-region families with sensitive babies to precisely target safe options and provide more scientific and effective nutritional solutions for sensitive babies.
By using the list, parents can conveniently screen nutritional supplements suitable for sensitive babies to reduce the risks caused by unknown allergens. For a long time, there are differences in allergen labeling in the industry. Witsbb's transparent list provides a new way of thinking and direction for allergen labeling in the industry, which to a certain extent, prompts the progress of the industry in allergen labeling management.
Witsbb has been committed to providing safe nutritional products for sensitive babies. It has witnessed it's development from being free from 8 common allergens at stage 1.0, to being free from 29 common food allergens at stage 2.0, and then to being free from 57 common food allergens at stage 3.0. Now, with the release of the Transparent List, Witsbb has officially entered the 4.0 stage.
"We understand that when choosing nutritional supplements for families with sensitive babies, it is important to avoid the risk of allergies while ensuring nutritional effectiveness," said the relevant person in charge of Witsbb, "the release of the 'Allergen Free 100+ Transparent List' is precisely to enable parents to save their hearts and babies to feel at ease with more transparent information." At present, the standardized development of the infant nutrition market is still under exploration. The practice of Witsbb can potentially provide a feasible reference model for the industry.
Witsbb "Allergy-free 100+ transparent list" Transparent Exclusion List:
https://www.witsbb.com.au/post/witsbb-allergen-free-100-transparent-exclusion-list
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

Witsbb Sets New Benchmark with Allergen-free 100+ Transparent List, Driving the Industry Standardization
|
Accomplished medical research leader will oversee NCCN programs to advance clinical trials and improve the quality, effectiveness, and accessibility of cancer care.
PLYMOUTH MEETING, Pa., April 9, 2026 /PRNewswire/ -- Today, the National Comprehensive Cancer Network® (NCCN®)—an alliance of leading cancer centers devoted to patient care, research, and education—announced the selection of Nancy L. Lewis, MD, MBS, FACP, as new Chief Scientific Officer (CSO).
Dr. Lewis is an experienced biomedical researcher with expertise in clinical trials for treating solid tumors and hematologic malignancies. She most recently served as Senior Clinical Program Leader for Novartis Pharmaceuticals, after several years as an Associate Professor for various prestigious academic cancer centers. She holds degrees from Penn State University, Temple University School of Medicine, and Rutgers University. Dr. Lewis completed residency at the University of Rochester and Fellowship at Fox Chase Cancer Center—one of NCCN's founding Member Institutions.
"This is a pivotal time for cancer research; there are many successes to build on yet so much still to do. Dr. Lewis is the perfect person to oversee the work we do to foster innovation and knowledge discovery that improves the lives of people with cancer," said Crystal S. Denlinger, MD, CEO, NCCN. "She will be an important asset to our leadership team as she helps us ensure our research and clinical programs continue to advance outcomes in oncology around the world."
Dr. Lewis has previously been honored with awards from some of the foremost cancer organizations across the country, including the American Association for Cancer Research (AACR), The American Society of Clinical Oncology (ASCO), and the American Cancer Society (ACS).
As CSO, Dr. Lewis will assist in the NCCN Oncology Research Program (ORP) as well as key aspects of the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) and NCCN Guidelines for Patients®. She will have oversight of the NCCN Biomarkers Compendium®; NCCN Radiation Therapy Compendium™, and NCCN Imaging Appropriate Use Criteria™. Dr. Lewis will also assist with NCCN's Continuing Medical Education Program and with relevant global and policy initiatives.
"I am thrilled to join the National Comprehensive Cancer Network and contribute to its extraordinary legacy of advancing high‑quality cancer care," said Dr. Lewis. "I look forward to working with NCCN's exceptional collaborators to tackle emerging challenges and drive innovative solutions that improve the lives of patients everywhere."
Dr. Lewis will succeed Dr. Denlinger who previously held the position of CSO before becoming CEO for the organization. Dr. Lewis will assume her new role in May 2026.
About the National Comprehensive Cancer Network
The National Comprehensive Cancer Network® (NCCN®) is a not-for-profit alliance of leading cancer centers devoted to patient care, research, and education. NCCN is dedicated to defining and advancing quality, effective, equitable, and accessible cancer care and prevention so all people can live better lives. The NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) provide transparent, evidence-based, expert consensus-driven recommendations for cancer treatment, prevention, and supportive services; they are the recognized standard for clinical direction and policy in cancer management and the most thorough and frequently-updated clinical practice guidelines available in any area of medicine. The NCCN Guidelines for Patients® provide expert cancer treatment information to inform and empower patients and caregivers, through support from the NCCN Foundation®. NCCN also advances continuing education, global initiatives, policy, and research collaboration and publication in oncology. Visit NCCN.org for more information.
Media Contact:
Rachel Darwin
267-622-6624
darwin@nccn.org
Accomplished medical research leader will oversee NCCN programs to advance clinical trials and improve the quality, effectiveness, and accessibility of cancer care.
PLYMOUTH MEETING, Pa., April 9, 2026 /PRNewswire/ -- Today, the National Comprehensive Cancer Network® (NCCN®)—an alliance of leading cancer centers devoted to patient care, research, and education—announced the selection of Nancy L. Lewis, MD, MBS, FACP, as new Chief Scientific Officer (CSO).
Dr. Lewis is an experienced biomedical researcher with expertise in clinical trials for treating solid tumors and hematologic malignancies. She most recently served as Senior Clinical Program Leader for Novartis Pharmaceuticals, after several years as an Associate Professor for various prestigious academic cancer centers. She holds degrees from Penn State University, Temple University School of Medicine, and Rutgers University. Dr. Lewis completed residency at the University of Rochester and Fellowship at Fox Chase Cancer Center—one of NCCN's founding Member Institutions.
"This is a pivotal time for cancer research; there are many successes to build on yet so much still to do. Dr. Lewis is the perfect person to oversee the work we do to foster innovation and knowledge discovery that improves the lives of people with cancer," said Crystal S. Denlinger, MD, CEO, NCCN. "She will be an important asset to our leadership team as she helps us ensure our research and clinical programs continue to advance outcomes in oncology around the world."
Dr. Lewis has previously been honored with awards from some of the foremost cancer organizations across the country, including the American Association for Cancer Research (AACR), The American Society of Clinical Oncology (ASCO), and the American Cancer Society (ACS).
As CSO, Dr. Lewis will assist in the NCCN Oncology Research Program (ORP) as well as key aspects of the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) and NCCN Guidelines for Patients®. She will have oversight of the NCCN Biomarkers Compendium®; NCCN Radiation Therapy Compendium™, and NCCN Imaging Appropriate Use Criteria™. Dr. Lewis will also assist with NCCN's Continuing Medical Education Program and with relevant global and policy initiatives.
"I am thrilled to join the National Comprehensive Cancer Network and contribute to its extraordinary legacy of advancing high‑quality cancer care," said Dr. Lewis. "I look forward to working with NCCN's exceptional collaborators to tackle emerging challenges and drive innovative solutions that improve the lives of patients everywhere."
Dr. Lewis will succeed Dr. Denlinger who previously held the position of CSO before becoming CEO for the organization. Dr. Lewis will assume her new role in May 2026.
About the National Comprehensive Cancer Network
The National Comprehensive Cancer Network® (NCCN®) is a not-for-profit alliance of leading cancer centers devoted to patient care, research, and education. NCCN is dedicated to defining and advancing quality, effective, equitable, and accessible cancer care and prevention so all people can live better lives. The NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) provide transparent, evidence-based, expert consensus-driven recommendations for cancer treatment, prevention, and supportive services; they are the recognized standard for clinical direction and policy in cancer management and the most thorough and frequently-updated clinical practice guidelines available in any area of medicine. The NCCN Guidelines for Patients® provide expert cancer treatment information to inform and empower patients and caregivers, through support from the NCCN Foundation®. NCCN also advances continuing education, global initiatives, policy, and research collaboration and publication in oncology. Visit NCCN.org for more information.
Media Contact:
Rachel Darwin
267-622-6624
darwin@nccn.org
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

Nancy L. Lewis, MD, MBS, FACP, Named New Chief Scientific Officer for National Comprehensive Cancer Network (NCCN)