|
Findings presented at the International VT Symposium and published simultaneously in Circulation.
CARDIFF-BY-THE-SEA, Calif. and PHILADELPHIA, Oct. 11, 2025 /PRNewswire/ -- Field Medical, Inc. today announced that Circulation has published six-month outcomes from the Ventricular Catheter Ablation Study (VCAS), the first-in-human evaluation of its FieldForce™ Ablation System for ventricular tachycardia (VT). Results were also presented as a late-breaking trial at the 20th Annual International Symposium on Ventricular Arrhythmias (VT Symposium).
Results at Six Months
VCAS is a prospective, multicenter feasibility trial evaluating the safety and performance of the FieldForce Ablation System in patients with VT. Unlike conventional approaches, the system delivers a proprietary high-voltage, short-pulse waveform designed to penetrate dense scar tissue while minimizing thermal injury.
Key Findings:
- 82% freedom from recurrent VT/VF or ICD therapy
- 98% reduction in VT/VF burden (episodes)
- 11.5% had a primary safety event with 0 therapy-related complications
"While this remains an initial feasibility study, the six-month outcomes are highly encouraging. Achieving 82% freedom from recurrence and a 98% reduction in arrhythmia burden with a nonthermal, tissue-selective energy is a meaningful result in VT therapy. Importantly, this is the first time we've seen evidence that PFA can reach deep, transmural scar tissue in the ventricle, a long-standing challenge with existing energy sources," said Vivek Reddy, M.D., co-principal investigator, lead author, and electrophysiologist at Mount Sinai, New York. "These findings give me cautious optimism that with continued refinement, this approach could represent an important advance in the treatment of scar-related VT."
With U.S. Food and Drug Administration's (FDA) Breakthrough Device designation and acceptance into the FDA Total Product Life Cycle (TAP) Pilot Program, Field Medical is advancing this program toward a pivotal trial and a rigorous evaluation of high-voltage focal PFA in VT.
"It is rare for initial feasibility data to be published in Circulation, and this underscores both the rigor and the significance of the work," said Steven Mickelsen, M.D., founder and chief technology officer of Field Medical. "Our mission has always been to unite scientific credibility with innovation. These findings mark an important milestone as we continue to evaluate pulsed field ablation for its potential to improve outcomes for patients with ventricular arrhythmias."
Looking ahead, the company is evaluating additional applications of its FieldForce Ablation System beyond VT and expects to present initial feasibility findings in atrial fibrillation (AF) at a major scientific meeting in early 2026.
About FieldForce™ Ablation System
The FieldForce Ablation System features a single-point contact force PFA catheter with an innovative design utilizing proprietary FieldBending™ technology to deliver targeted, brief, high-intensity electric fields. This next-generation PFA technology was designed to deliver both precise targeted lesions and large volume transmural lesions in the ventricle.
About Field Medical®, Inc.
Founded in 2022, Field Medical is a clinical-stage medical technology company committed to advancing pulsed field ablation (PFA) solutions for complex cardiac arrhythmias. Its FieldForce Ablation System integrates a focal catheter design with proprietary FieldBending energy designed to safely deliver efficient, precise ablation with the goal of improving outcomes in ventricular and atrial arrhythmia treatment. In 2024, Field Medical earned Breakthrough Device Designation and gained entry into the FDA TAP Pilot Program for its ventricular tachycardia indication.
For more information, visit www.fieldmedicalinc.com and follow us on LinkedIn X, and YouTube.
The FieldForce™ Ablation System is an investigational device and is limited by federal (or United States) law to investigational use.
Source:
Reddy VY, et al. High-Voltage Focal Pulsed Field Ablation to Treat Scar-Related Ventricular Tachycardia: The First-in-Human VCAS Trial. Circulation. Published online ahead of print October 10, 2025. doi:10.1161/CIRCULATIONAHA.125.077025
CONTACT: Holly Windler, 619.929.1275, holly.windler@gmail.com
Photo - https://mma.prnasia.com/media2/2792975/Field_Medical_FieldForce__Ablation_System_Next_generation_PFA.jpg?p=medium600
Logo - https://mma.prnasia.com/media2/2574173/Field_Medical_Logo_Block_WhiteOnBlack_Logo.jpg?p=medium600
Findings presented at the International VT Symposium and published simultaneously in Circulation.
CARDIFF-BY-THE-SEA, Calif. and PHILADELPHIA, Oct. 11, 2025 /PRNewswire/ -- Field Medical, Inc. today announced that Circulation has published six-month outcomes from the Ventricular Catheter Ablation Study (VCAS), the first-in-human evaluation of its FieldForce™ Ablation System for ventricular tachycardia (VT). Results were also presented as a late-breaking trial at the 20th Annual International Symposium on Ventricular Arrhythmias (VT Symposium).
Results at Six Months
VCAS is a prospective, multicenter feasibility trial evaluating the safety and performance of the FieldForce Ablation System in patients with VT. Unlike conventional approaches, the system delivers a proprietary high-voltage, short-pulse waveform designed to penetrate dense scar tissue while minimizing thermal injury.
Key Findings:
- 82% freedom from recurrent VT/VF or ICD therapy
- 98% reduction in VT/VF burden (episodes)
- 11.5% had a primary safety event with 0 therapy-related complications
"While this remains an initial feasibility study, the six-month outcomes are highly encouraging. Achieving 82% freedom from recurrence and a 98% reduction in arrhythmia burden with a nonthermal, tissue-selective energy is a meaningful result in VT therapy. Importantly, this is the first time we've seen evidence that PFA can reach deep, transmural scar tissue in the ventricle, a long-standing challenge with existing energy sources," said Vivek Reddy, M.D., co-principal investigator, lead author, and electrophysiologist at Mount Sinai, New York. "These findings give me cautious optimism that with continued refinement, this approach could represent an important advance in the treatment of scar-related VT."
With U.S. Food and Drug Administration's (FDA) Breakthrough Device designation and acceptance into the FDA Total Product Life Cycle (TAP) Pilot Program, Field Medical is advancing this program toward a pivotal trial and a rigorous evaluation of high-voltage focal PFA in VT.
"It is rare for initial feasibility data to be published in Circulation, and this underscores both the rigor and the significance of the work," said Steven Mickelsen, M.D., founder and chief technology officer of Field Medical. "Our mission has always been to unite scientific credibility with innovation. These findings mark an important milestone as we continue to evaluate pulsed field ablation for its potential to improve outcomes for patients with ventricular arrhythmias."
Looking ahead, the company is evaluating additional applications of its FieldForce Ablation System beyond VT and expects to present initial feasibility findings in atrial fibrillation (AF) at a major scientific meeting in early 2026.
About FieldForce™ Ablation System
The FieldForce Ablation System features a single-point contact force PFA catheter with an innovative design utilizing proprietary FieldBending™ technology to deliver targeted, brief, high-intensity electric fields. This next-generation PFA technology was designed to deliver both precise targeted lesions and large volume transmural lesions in the ventricle.
About Field Medical®, Inc.
Founded in 2022, Field Medical is a clinical-stage medical technology company committed to advancing pulsed field ablation (PFA) solutions for complex cardiac arrhythmias. Its FieldForce Ablation System integrates a focal catheter design with proprietary FieldBending energy designed to safely deliver efficient, precise ablation with the goal of improving outcomes in ventricular and atrial arrhythmia treatment. In 2024, Field Medical earned Breakthrough Device Designation and gained entry into the FDA TAP Pilot Program for its ventricular tachycardia indication.
For more information, visit www.fieldmedicalinc.com and follow us on LinkedIn X, and YouTube.
The FieldForce™ Ablation System is an investigational device and is limited by federal (or United States) law to investigational use.
Source:
Reddy VY, et al. High-Voltage Focal Pulsed Field Ablation to Treat Scar-Related Ventricular Tachycardia: The First-in-Human VCAS Trial. Circulation. Published online ahead of print October 10, 2025. doi:10.1161/CIRCULATIONAHA.125.077025
CONTACT: Holly Windler, 619.929.1275, holly.windler@gmail.com
Photo - https://mma.prnasia.com/media2/2792975/Field_Medical_FieldForce__Ablation_System_Next_generation_PFA.jpg?p=medium600
Logo - https://mma.prnasia.com/media2/2574173/Field_Medical_Logo_Block_WhiteOnBlack_Logo.jpg?p=medium600
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

Circulation Publishes 6-Month Outcomes from First-in-Human VCAS Trial of FieldForce™ Pulsed Field Ablation System: 82% Freedom from VT/VF Recurrence and 98% Reduction in Burden
World's Leading Provider of Cutting-Edge Smart Ring Solutions
NEW YORK, May 10, 2026 /PRNewswire/ -- RingConn opened Gen 3 pre-orders on May 5th ahead of its official May 29th launch, marking the occasion with a feature on the iconic One Times Square billboard in New York City on the first day of pre-orders. The next-generation device combines holistic health tracking, subtle smart alerts, and new premium finishes, delivering an ultra-lightweight and slim design without compromising long battery life. To celebrate the launch, customers who pre-order through the official RingConn website can enjoy a limited-time 10% discount.*
Vascular Trend tracking
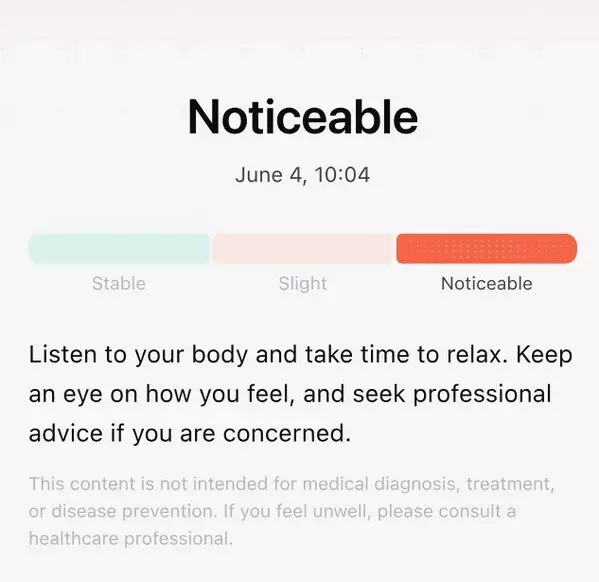
RingConn Gen 3 Smart Ring introduces advanced vascular insights, designed to help users observe long‑term vascular trends through continuous data collection.*
Built on over a decade of research in vascular algorithms and modeling, RingConn's vascular capability moves beyond single-point measurements to long-term trend-based assessment. The system requires periodic user calibration to ensure improved accuracy, with recommendations for calibration three times within the first 24 hours and monthly thereafter. By combining user-input blood pressure data with ring-derived vascular load indicators, the system provides a more comprehensive view of overall vascular health.
Once calibrated, Gen 3 enables automatic vascular load assessments during periods of sleep or low motion, allowing users to continuously track meaningful changes over time without frequent manual input. The system also incorporates key influencing factors—including circadian rhythm adaptation, sleep respiratory health, post-exercise vascular recovery, and overall daily vascular stability—to help users better understand the relationship between lifestyle and cardiovascular patterns.
Effortless 24/7 Wearability with Extended Battery Life
RingConn Gen 3 Smart Ring is designed for continuous and comfortable wear, with a fully upgraded experience centered on durability and long-term usability. Featuring a refined ultra-lightweight form factor and enhanced engineering design, the device is built to seamlessly integrate into daily life, enabling consistent and uninterrupted health data tracking.
Powered by RingConn EcoPower Tech 2.0, the Gen 3 features an upgraded battery system and improved energy density, delivering up to 11–14 days of battery life in standard usage without vibration mode. This reduces charging frequency and supports continuous, long-term health monitoring with minimal interruption.
The ring adopts RingConn's proprietary ergonomic design, resulting in a lighter and thinner profile that conforms more naturally to the finger. With IP68 water resistance up to 100 meters, the Gen 3 is designed for durability in everyday use, enabling reliable performance across a wide range of real-life scenarios. Together, these features allow users to wear the device seamlessly through typing, handwashing, showering, swimming, and sleep - ensuring uninterrupted physiological data collection day and night.
A refined brushed finish, achieved through an advanced laser-engraving process, enhances the device's surface texture with greater precision and depth. The upgraded finishing process requires over 30% more production time compared to conventional methods, reflecting a higher level of craftsmanship. Expanded color options further enhance versatility, allowing the ring to blend naturally into both casual and professional environments while encouraging consistent daily wear.
Holistic Health Tracking
RingConn Gen 3 Smart Ring delivers comprehensive health tracking across multiple key dimensions, including sleep, sleep apnea, heart rate, blood oxygen, activity, stress, and women's health management such as menstrual cycle tracking and pregnancy preparation-related insights. Through continuous monitoring and long-term analysis, the device provides users with a more complete understanding of their health status over time, supporting more informed daily lifestyle decisions.
The system is powered by an advanced VCSEL infrared sensing solution, designed to enhance data stability and accuracy across a wide range of wearing conditions and real-world scenarios, strengthening the foundation for long-term health trend analysis. In internal testing, the system achieved a resting heart rate accuracy of ≥98.58%, a blood oxygen mean absolute error (MAE) of ≤0.95%, a step count accuracy of ≥97.20%, and a temperature accuracy of ±0.08°C, demonstrating strong performance in multi-dimensional health data capture.
Subtle Smart Alert
RingConn Gen 3 Smart Ring introduces subtle smart alerts to provide timely feedback when key health changes are detected. Through gentle vibration-based notifications, the device can also prompt users in scenarios such as prolonged inactivity or low battery, helping support more balanced daily usage and awareness. In quiet environments — such as during meetings or conversations — these silent notifications allow users to stay informed without disruptive sounds or attention-grabbing screen notifications.
The alert system is fully customizable, allowing users to enable or disable notifications based on personal preference. By prioritizing minimal disruption and silent vibration cues over intrusive alerts, RingConn ensures that health reminders remain discreet, context-aware, and seamlessly integrated into everyday life.
*Pre-order offer: The 10% limited-time discount is available exclusively at the RingConn official website from May 5 through June 10, 2026.
*This product is not intended to diagnose, treat, cure, or prevent any disease or medical condition. As a pre-diagnostic tool, the Gen 3 does not replace the diagnosis or treatment by a qualified healthcare professional.
For more information or to place a pre-order, visit:
https://bit.ly/ringconngen3
Follow RingConn on social media:
Instagram: https://bit.ly/ringconninstagram
TikTok: https://bit.ly/ringconntiktok
About RingConn
Established in 2021, RingConn is a user-centric company, driven by continuous innovation, to provide professional, continuous, and thoughtful health monitoring products and services. RingConn is dedicated to becoming users' most trusted guardian for their physical and mental well-being.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

RingConn Gen 3 Smart Ring Opens for Pre-Order: Bringing Vascular Insights to Effortless Everyday Wear
RingConn Gen 3 Smart Ring Opens for Pre-Order: Bringing Vascular Insights to Effortless Everyday Wear

RingConn Gen 3 Smart Ring Opens for Pre-Order: Bringing Vascular Insights to Effortless Everyday Wear