Accelerates growth with new signings in Seoul and Busan
SINGAPORE, Jan. 13, 2026 /PRNewswire/ -- Wyndham Hotels & Resorts is stepping up its expansion in South Korea, opening its first managed hotel and adding two new development projects as it seeks to build scale in a market rebounding faster than much of Northeast Asia.
The company recently opened the 529-room Wyndham Goseong Gangwon, near Seoraksan National Park, and has signed agreements for Howard Johnson by Wyndham Seoul, expected to open in 2028, and Wyndham Busan, slated for 2030.
Wyndham's expansion comes as South Korea's tourism sector continues to rebound, supported by resilient domestic travel and a steady recovery in international arrivals. According to the Korea Tourism Organization, the country recorded 15.821 million inbound visitors between January and October, a 15.2% increase from a year earlier. Industry executives expect international demand to continue improving into 2025, supporting increased investment in branded accommodation across urban and resort markets.
"South Korea's booming tourism market presents a compelling opportunity for Wyndham, which is why we're focused on strategic expansion and strengthening our presence through high-caliber additions like Wyndham Goseong, Gangwon. With global scale, an industry-leading loyalty platform, and deep local expertise, Wyndham has a proven record of helping owners not only drive performance, but generate long-term value while delivering exceptional guest experiences."
— Ben Schumacher, Regional Vice President, Operations, APAC, Wyndham Hotels & Resorts
Wyndham Goseong, Gangwon: A Coastal Retreat for Relaxation and Events
The opening of Wyndham Goseong, Gangwon marks Wyndham's first managed hotel in South Korea and a key milestone in the Company's local operations. Located adjacent to Bongpo Beach and Seoraksan National Park, the 529-room, ocean-view property is designed to serve leisure travelers, families, and MICE groups.
Over the past two years, Wyndham has expanded its footprint in South Korea through new brands and operating models, including the introduction of Wyndham Grand in 2023 and more recently, Trademark Collection by Wyndham in 2024. These additions build on an established portfolio of 30 hotels with more than 9,200 rooms across brands such as Days Inn®, Howard Johnson®, Ramada Plaza®, Ramada®, Ramada Encore®, Ramada Hotel & Suites®, Wyndham Grand® and Trademark Collection®, enabling Wyndham to serve diverse traveler segments while supporting owners with scalable, globally recognized brands.
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

Wyndham Enhances Position in South Korea with First Managed Hotel Opening
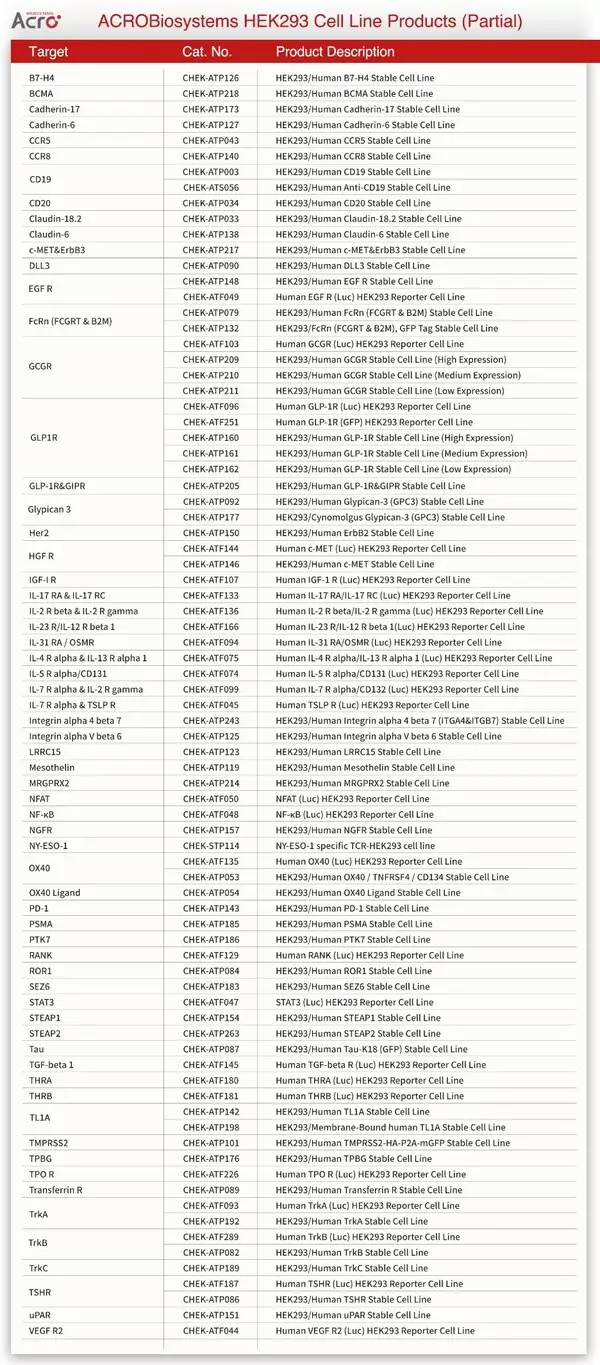
NEWARK, Del., May 10, 2026 /PRNewswire/ -- ACROBiosystems has implemented a major upgrade to its global licensing framework for HEK293 functional cell lines, transitioning from a complex administrative process to a streamlined, purchase-based authorization model. This policy shift addresses the industry-wide challenge of compliance delays by embedding intellectual property clearance directly into the procurement process, thereby empowering global biopharmaceutical innovation with a more efficient and barrier-free R&D experience.
The official statement reads as follows:
The innovative development of biopharmaceuticals relies on stable, legally compliant R&D tools. Functional cell lines serve as a versatile vital engine driving full-cycle breakthroughs in innovative drug R&D, and the ease of obtaining usage authorization for functional cell lines directly impacts projects timelines. To simplify compliance arrangements and reduce administrative burdens for global partners, ACROBiosystems has upgraded its global permitted use policy for HEK293 functional cell line products. The updated framework provides an expansive scope of usage rights to encompass not only internal research but also certain activities from early discovery well into clinical development. It provides clear and standardized legitimate usage scopes and removes redundant review steps, enabling researchers to conduct R&D activities in full compliance with IP and contractual requirements. Customers operating within the defined permitted use scope are now exempt from additional license processing and associated cost, allowing them to focus on core drug R&D and accelerate project progress, with greatly improved efficiency.
- Core Policy & Compliance Framework
The upgraded policy simplifies compliance processes while establishing clear legitimate usage boundaries. Customers operating within the defined permitted use scope below are entitled to utilize HEK293 functional cell line products for specified activities without additional application or approval.
1.Permitted Usage Scope
HEK293 functional cell line products are authorized for use in internal research, drug discovery, assay development, internal quality assurance (QA) testing and lot release analysis.
*Note: For all other commercial uses beyond the above scopes, customers shall contact ACROBiosystems to obtain a license through flexible options.
2.Regional & Usage Notes
This policy applies to all global markets except the Greater China region, delivering seamless, efficient access to the HEK293 functional cell line products for research and development activities. By eliminating unnecessary administrative hurdles, this streamlined compliance approach empowers global researchers to redirect their focus toward core R&D priorities—rather than navigating complex compliance procedures.
For users in the Greater China region, the HEK293 functional cell line products are permitted for research use only, with separate official authorization required for any extended usage needs.
With localized teams across the United States, Europe and the Asia-Pacific area, ACROBiosystems provides dedicated policy guidance and one-stop compliance consulting tailored to this HEK293 functional cell line global license upgrade. Our regional experts offer timely interpretation of updated usage rules, streamlined consultation for scope expansion requests, and professional technical backing, helping global customers quickly adapt to the new policy and carry out functional cell line-related R&D in a fully compliant, efficient manner.
This policy upgrade further clarifies global permitted-use terms for HEK293 functional cell line products, streamlines daily compliance management, and lowers overall operational and compliance costs for global drug developers. By establishing a clearer, more flexible global compliance framework, ACROBiosystems enables the standardized, orderly and scalable expansion of HEK293 functional cell line compliant applications across diverse R&D scenarios. Moving forward, we will continue to optimize global IP governance and licensing mechanisms, deliver regulated, transparent and user-friendly functional cell line solutions, and support customers in exploring broader preclinical and translational research possibilities with fully compliant functional cell line resources, jointly accelerating high-quality innovative drug development worldwide.
About ACROBiosystems Group
ACROBiosystems Group, founded in 2010 and listed in 2021, is a biotechnology company aimed at being a cornerstone of the global biopharmaceutical and healthcare industries by providing innovative products and business models. The company spans across the globe and maintains offices, R&D centers, and production bases in more than 15 different cities within the United States, Switzerland, the United Kingdom and Germany. ACROBiosystems Group has established numerous long-term and stable partnerships with the world's top pharmaceutical enterprises, including Pfizer, Novartis, and Johnson & Johnson, and numerous well-known academic institutes. The company comprises several subsidiaries such as ACROBiosystems, bioSeedin, Condense Capital, and ACRODiagnostics.
ACROBiosystems' brands include Resilient Supply, CytoPak, SAFENSURE, FLAG, Star Staining, Aneuro, ComboX, GENPower and many others. Its main products and services are recombinant proteins, kits, antibodies, scientific services, and other related products. ACROBiosystems employs a strict quality control system for its products that are used in biopharmaceutical research and development, production, and clinical application. This includes targeted discovery and validation, candidate drug screening/optimization, CMC development and pilot production, preclinical research, clinical trials, commercial production, and clinical application of companion diagnostics.
Through the continuous development of new technologies and products, ACROBiosystems Group creates value for the global pharmaceutical industry and actively empowers our partners. The company is dedicated to accelerating the drug development process, including targeted therapies, immunotherapeutic drugs, and their clinical applications, and contributes to global health.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
ACROBiosystems Launches Global License Solution Upgrade for HEK293 Functional Cell Lines, Streamlining Compliance to Accelerate Biopharmaceutical R&D











