VANCOUVER, BC, Feb. 10, 2026 /PRNewswire/ -- WAT Medical is proud to announce the publication of a landmark randomized clinical trial in JAMA Surgery, one of the world's most reputable and high-impact surgical journals. The study confirms that the EmeTerm wristband — a wearable device utilizing transcutaneous acupoint electrical stimulation — provides significantly better control of moderate to severe postoperative nausea and vomiting (PONV) compared to metoclopramide, a widely used antiemetic medication.
The patient-blinded, multicenter randomized clinical trial enrolled 232 female patients who developed moderate to severe PONV (numerical rating score [NRS] ≥4) following thyroidectomy or anterior cervical surgery under general anesthesia. Conducted across four leading hospitals, participants were randomized 1:1 to receive either active transcutaneous electrical acupoint stimulation (TEAS) via the EmeTerm wristband targeting the median nerve at acupoint PC6, or a control intervention consisting of a sham device combined with intravenous metoclopramide.
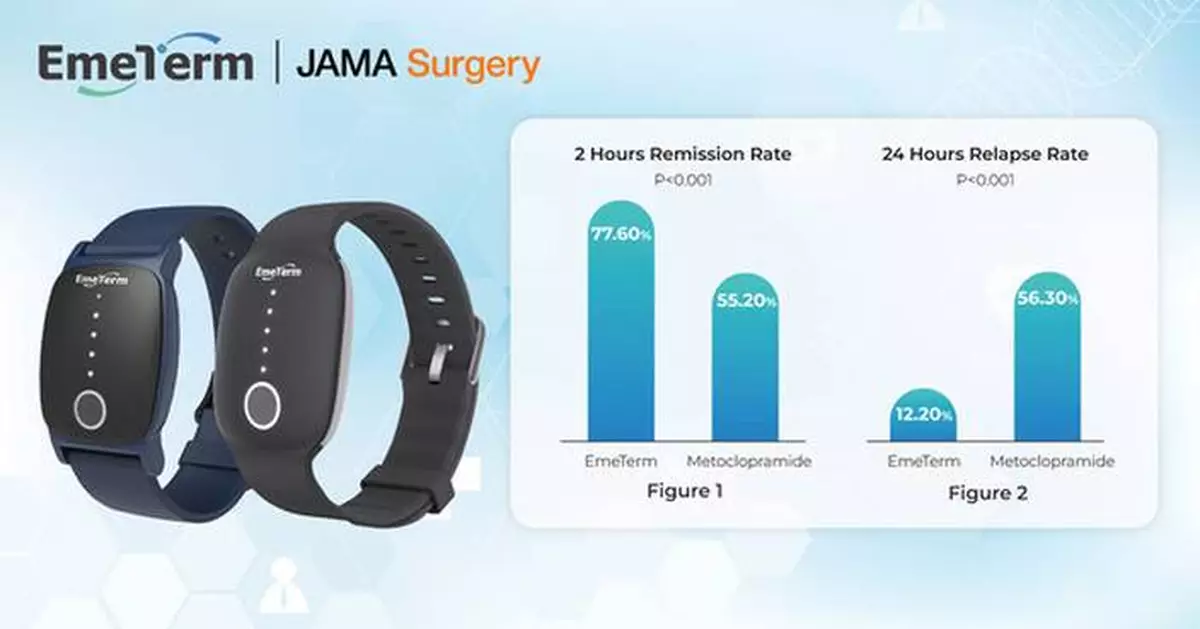
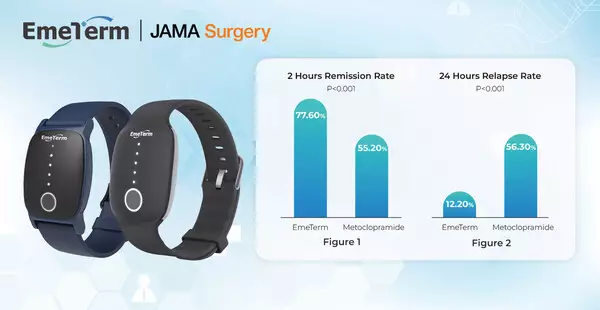
The primary endpoint was the 2-hour remission rate of PONV, while secondary endpoints included 24-hour relapse rates and crossover responses.
Key Findings
- 2-Hour Remission Rate: EmeTerm significantly improved PONV remission compared with metoclopramide (77.6% vs 55.2%, P < 0.001) (Figure 1).
- 24-Hour Relapse Rate: Patients treated with EmeTerm experienced significantly lower relapse rates than those treated with metoclopramide (12.2% vs 56.3%, P < 0.001) (Figure 2).
- Safety: No device-related serious adverse events were reported.
These findings demonstrate that EmeTerm not only achieves superior and sustained symptom control compared with metoclopramide, but also offers a favorable safety and tolerability profile. The results highlight a shift toward non-pharmacological, patient-centered approaches in perioperative care — enhancing patient autonomy, minimizing drug-related side effects, and improving overall recovery outcomes.
Developed by WAT Medical Enterprise Ltd., EmeTerm represents the company's commitment to evidence-based innovation in neuromodulation for nausea management. The device has received multiple international regulatory clearances, including approvals from the U.S. FDA, Health Canada, and Australia's TGA, affirming compliance with global safety and quality standards.
Publication in JAMA Surgery underscores the rigor and impact of the study, validating EmeTerm as an effective, non-drug therapeutic option for managing active PONV. With a growing body of clinical evidence, EmeTerm is being increasingly adopted by clinicians and hospitals worldwide as a reliable solution for nausea control in perioperative settings.
References
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **
WAT Medical's EmeTerm Wearable Device Demonstrates Superior Control of Moderate to Severe Postoperative Nausea and Vomiting in JAMA Surgery-Published Clinical Trial