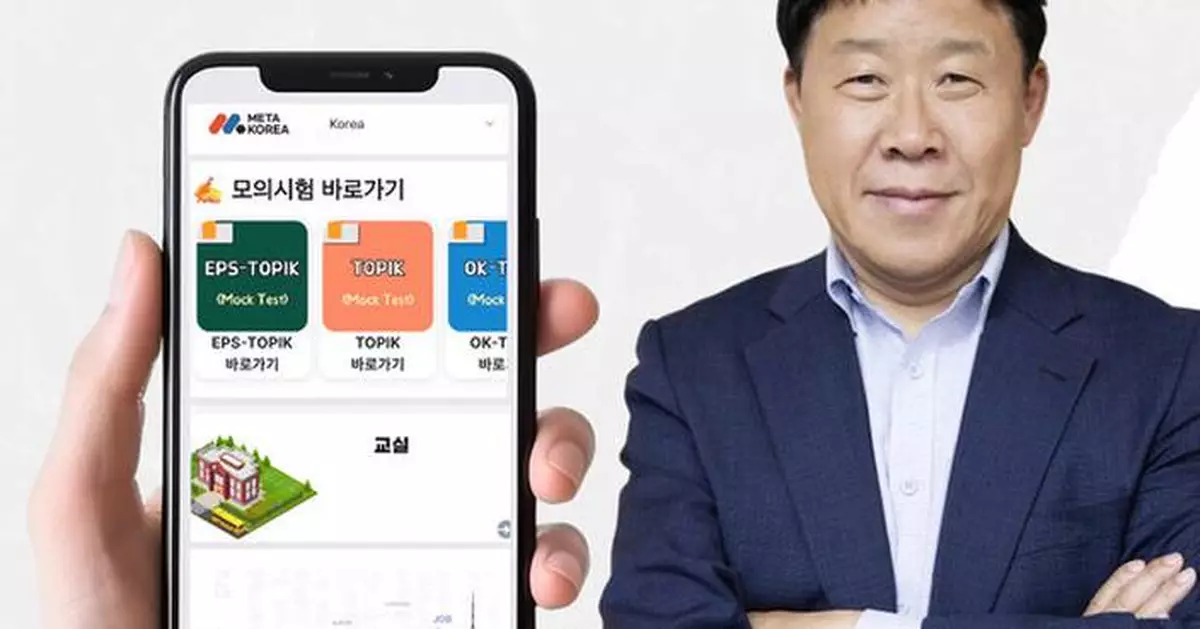
SEOUL, South Korea, Feb. 26, 2026 /PRNewswire/ -- As K-culture continues to capture worldwide attention, the number of foreigners seeking to learn Korean has surged. However, many regions still lack adequate educational institutions. Against this backdrop, Han Il-hwan, CEO of Han Edu Tech Co., Ltd., is gaining recognition for developing MetaKorea, an application that delivers Korean language education and testing content, enabling users around the world to learn Korean anytime, anywhere.
Han Edu Tech Develops "MetaKorea," a Global Platform for Certified Korean Exams and Education
Han Edu Tech envisions a world where access barriers to Korean language education and official exams such as TOPIK, EPS-TOPIK, and OK-TEST no longer exist, regardless of time or location. CEO Han Il-hwan explained, "We founded Han Edu Tech with the goal of widely promoting the Korean language. Drawing on years of experience in the education sector, we developed MetaKorea, a platform that allows users to access Korean language courses, official test information, and mock exams all within a single app." He added, "We hope international students will take an interest in Han Edu Tech's OK-TEST and actively use MetaKorea. We believe the app can help Korean language learners worldwide improve their business Korean skills and gain advantages in both domestic and international employment."
App Developed to Support Korean Language Globalization Through Testing and Education
Han emphasized that OK-TEST, a business Korean exam modeled after TOEIC, has become the third officially recognized foreign language test in China, following English TOEIC and Japanese J-TEST. It is the only Korean language test officially recognized by the Chinese government. Approved in 2010 by China's Ministry of Human Resources and Social Security as a national qualification exam, it is currently administered at over 300 Chinese universities and at Korean-invested companies in China, including POSCO, SK Hynix, and Woori Bank.
Following agreements signed in 2023 with Vietnam's VXT College, NEWTATCO (a state-owned enterprise under Vietnam's Ministry of National Defense), and FTC, Han Edu Tech also entered into an MOU in April 2024 with Vietnam's JHL Group to expand Korean language education initiatives. Beyond these markets, the company plans to expand into India, Indonesia, Nepal, Uzbekistan, North America, Europe, and South America—regions with strong demand for Korean language learning and significant Korean corporate presence. The OK-TEST administered by Han Edu Tech is designed for foreigners seeking to study or work in Korea, as well as for advanced learners with TOPIK Level 5 or higher who want to communicate more fluently with native speakers. The exam is also registered domestically in Korea as a private certification. Through the MetaKorea app, users can practice with past exam questions to prepare in advance.
MetaKorea's CEO Han Il-hwan
From 1994 to 2002, Han operated e-learning LMS solutions and educational websites, eventually listing them on the KOSDAQ stock market. In April 2017, he founded Han Edu Tech to promote the globalization of the Korean language through information and communication technology, focusing on the OK-TEST Occupational Korean Proficiency Test, related certification exams, and educational publishing to improve employment opportunities both in Korea and internationally.
Recently, the company as accelerated efforts to expand beyond China into Vietnam and other Asian countries, as well as North America and Europe. Han also established the China Business Academy in Beijing in 2002, where, until 2016, he ran the government-funded "K-Move School" overseas employment training program operated by Korea's Human Resources Development Service. During that time, he trained approximately 1,500 participants, successfully placing around 1,200 of them in Korean-invested companies across China.
For more information, please visit http://www.oktest.org
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

Han Edu Tech CEO Han Il-hwan Leads Global Expansion of K-Korean Language Testing Through the MetaKorea App
|
SEOUL, South Korea, Feb. 26, 2026 /PRNewswire/ -- CJ BIO announced the publication of clinical research findings on its postbiotic ingredient, BiomeNrich™ POST SZ075, in an international peer-reviewed journal. The study strengthens the scientific foundation of the ingredient and supports its positioning as a science-based inner beauty solution.
Growing Importance of Scientific Validation in the Inner Beauty Market
The global beauty and personal care market is rapidly shifting toward "Beauty from within" approaches that improve skin condition through internal health support. In particular, the gut-skin axis has emerged as a key biological pathway linking gut microbiota to skin health, driving increased research into probiotic and postbiotic-based ingestible skincare solutions.
At the same time, consumers increasingly seek objective and measurable evidence addressing visible skin concerns such as UV-induced photoaging, barrier impairment, dryness, roughness, and loss of elasticity. As a result, clinically validated biomarkers and instrumental assessments have become critical in substantiating functional ingredient claims.
CJ BIO's Science-Based Solution: BiomeNrich™ POST SZ075
BiomeNrich™ POST SZ075 is a postbiotic ingredient within CJ BIO's Condition-Specific biotics solution brand, BiomeNrich™. It is a 100% plant-based postbiotic produced by fermenting rice germ using Latilactobacillus sakei, a strain originally isolated from kimchi. The ingredient is positioned to support Sleep & Skin Health, developed based on the gut–skin axis concept for inner beauty and mental wellness applications.
The newly published study evaluated the ingredient's mechanisms of action in improving skin health and influencing skin-related biomarkers. Findings demonstrated significant modulation of key markers associated with barrier integrity, hydration, and wrinkle appearance, including reductions in wrinkle severity and transepidermal water loss (TEWL), alongside increases in skin elasticity and dermal density.
Dual-Action Efficacy of BiomeNrich™ POST SZ075 for Skin Health
A clinical study (n=111, 12 weeks, 500 mg/day) published in Food Science and Biotechnology (2025) confirms the dual functionality of BiomeNrich™ POST SZ075 in combating UV-induced wrinkles and enhancing hydration.
I. Skin wrinkle and roughness
Compared to the placebo, the test group showed significant resilience against UV-induced damage:
- 1,140% improvement in wrinkle parameters around the eyes.
- 614% increase in skin elasticity.
- 172% increase in dermal density
II. Skin hydration and Transepidermal Water Loss
The study also demonstrated improvements in hydration-related and barrier-related parameters:
- 1,872% decrease in Transepidermal Water Loss (TEWL)
- 497% improvement in skin roughness and 280% reduction in desquamation.
- 309% increase in skin moisture content.
Detailed research data on BiomeNrich™ POST SZ075 can be found in CJ BIO's Nutrition Insight article.
https://na2.hubs.ly/H03W1K20
Differentiation Through Integrated Evidence
CJ BIO established a multi-layered validation framework for BiomeNrich™ POST SZ075, encompassing in vitro, in vivo, and human clinical studies. This stepwise research design enables mechanistic verification alongside measurable clinical outcomes, strengthening scientific credibility. This integrated validation framework enables brand partners to reference both mechanistic insights and clinical findings when developing product concepts.
In addition to the published clinical study on skin health, CJ BIO has also conducted research exploring sleep-related benefits of BiomeNrich™ POST SZ075, supporting its potential relevance across multiple wellness applications. As a postbiotic ingredient, BiomeNrich™ POST SZ075 offers formulation stability advantages compared to live probiotics, which may facilitate broader product development possibilities.
CJ BIO stated, "This study provides scientific evidence that postbiotics can contribute to skin health. CJ BIO will continue expanding its portfolio of science-backed biotics and functional ingredients to deliver differentiated solutions in the inner beauty market."
About CJ BIO
CJ BIO is the biotechnology business unit of CJ CheilJedang, leveraging over 70 years of fermentation expertise to deliver functional ingredients and solutions for the food, taste, nutrition, and health industries. CJ BIO is committed to providing sustainable, science-validated bio-solutions that enhance quality of life for customers and consumers worldwide.
Contact Information
Media Inquiries:
CJ BIO
Email: cjbio_brand@cj.net
Website: https://na2.hubs.ly/H03W1JT0
SEOUL, South Korea, Feb. 26, 2026 /PRNewswire/ -- CJ BIO announced the publication of clinical research findings on its postbiotic ingredient, BiomeNrich™ POST SZ075, in an international peer-reviewed journal. The study strengthens the scientific foundation of the ingredient and supports its positioning as a science-based inner beauty solution.
Growing Importance of Scientific Validation in the Inner Beauty Market
The global beauty and personal care market is rapidly shifting toward "Beauty from within" approaches that improve skin condition through internal health support. In particular, the gut-skin axis has emerged as a key biological pathway linking gut microbiota to skin health, driving increased research into probiotic and postbiotic-based ingestible skincare solutions.
At the same time, consumers increasingly seek objective and measurable evidence addressing visible skin concerns such as UV-induced photoaging, barrier impairment, dryness, roughness, and loss of elasticity. As a result, clinically validated biomarkers and instrumental assessments have become critical in substantiating functional ingredient claims.
CJ BIO's Science-Based Solution: BiomeNrich™ POST SZ075
BiomeNrich™ POST SZ075 is a postbiotic ingredient within CJ BIO's Condition-Specific biotics solution brand, BiomeNrich™. It is a 100% plant-based postbiotic produced by fermenting rice germ using Latilactobacillus sakei, a strain originally isolated from kimchi. The ingredient is positioned to support Sleep & Skin Health, developed based on the gut–skin axis concept for inner beauty and mental wellness applications.
The newly published study evaluated the ingredient's mechanisms of action in improving skin health and influencing skin-related biomarkers. Findings demonstrated significant modulation of key markers associated with barrier integrity, hydration, and wrinkle appearance, including reductions in wrinkle severity and transepidermal water loss (TEWL), alongside increases in skin elasticity and dermal density.
Dual-Action Efficacy of BiomeNrich™ POST SZ075 for Skin Health
A clinical study (n=111, 12 weeks, 500 mg/day) published in Food Science and Biotechnology (2025) confirms the dual functionality of BiomeNrich™ POST SZ075 in combating UV-induced wrinkles and enhancing hydration.
I. Skin wrinkle and roughness
Compared to the placebo, the test group showed significant resilience against UV-induced damage:
- 1,140% improvement in wrinkle parameters around the eyes.
- 614% increase in skin elasticity.
- 172% increase in dermal density
II. Skin hydration and Transepidermal Water Loss
The study also demonstrated improvements in hydration-related and barrier-related parameters:
- 1,872% decrease in Transepidermal Water Loss (TEWL)
- 497% improvement in skin roughness and 280% reduction in desquamation.
- 309% increase in skin moisture content.
Detailed research data on BiomeNrich™ POST SZ075 can be found in CJ BIO's Nutrition Insight article.
https://na2.hubs.ly/H03W1K20
Differentiation Through Integrated Evidence
CJ BIO established a multi-layered validation framework for BiomeNrich™ POST SZ075, encompassing in vitro, in vivo, and human clinical studies. This stepwise research design enables mechanistic verification alongside measurable clinical outcomes, strengthening scientific credibility. This integrated validation framework enables brand partners to reference both mechanistic insights and clinical findings when developing product concepts.
In addition to the published clinical study on skin health, CJ BIO has also conducted research exploring sleep-related benefits of BiomeNrich™ POST SZ075, supporting its potential relevance across multiple wellness applications. As a postbiotic ingredient, BiomeNrich™ POST SZ075 offers formulation stability advantages compared to live probiotics, which may facilitate broader product development possibilities.
CJ BIO stated, "This study provides scientific evidence that postbiotics can contribute to skin health. CJ BIO will continue expanding its portfolio of science-backed biotics and functional ingredients to deliver differentiated solutions in the inner beauty market."
About CJ BIO
CJ BIO is the biotechnology business unit of CJ CheilJedang, leveraging over 70 years of fermentation expertise to deliver functional ingredients and solutions for the food, taste, nutrition, and health industries. CJ BIO is committed to providing sustainable, science-validated bio-solutions that enhance quality of life for customers and consumers worldwide.
Contact Information
Media Inquiries:
CJ BIO
Email: cjbio_brand@cj.net
Website: https://na2.hubs.ly/H03W1JT0
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

CJ BIO Publishes Clinical Study on BiomeNrich™ POST SZ075, a Postbiotic Ingredient Demonstrating Skin Improvement Benefits