SUZHOU, China, Feb. 27, 2026 /PRNewswire/ -- Basecare Medical (02170.HK) has recently encountered a key catalyst——its Gems one-step embryo culture medium has officially obtained the China Class III Medical Device Registration Certificate (National Medical Products Administration Registration No. 20253180071). This represents a core signal of the company's strategic transformation implementation and accelerated domestic substitution layout, and is expected to become an important driver for its valuation repair, warranting close attention.
From a market logic perspective, the approval of this product essentially serves as Basecare's "entry ticket" into the vast blue ocean of the multi-billion-yuan assisted reproductive consumables market. The underlying product strength and market foundation determine the certainty of its subsequent performance delivery, which serves as the core starting point for our analysis of its investment value.
I. Product Strength: Backed by Internationally Mature Assets, Market Competitiveness Need Not Be Overly Concerning
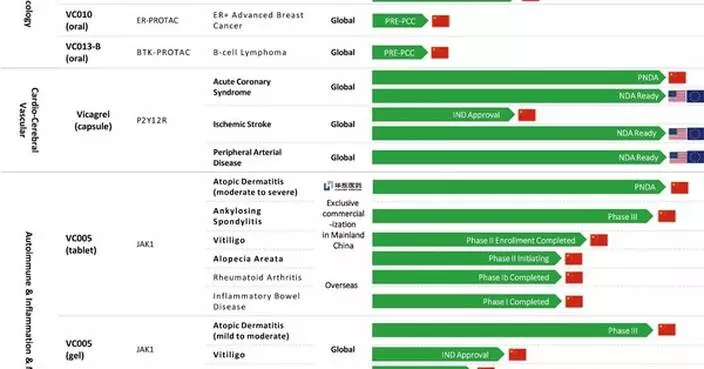
Unlike the model of many domestic enterprises involving "R&D from scratch and long-term investment," Basecare's Gems culture medium was acquired as an internationally mature asset – a particularly crucial point. Its technology originates from world-leading assisted reproductive institutions, was developed under the guidance of the "Father of Culture Media," has undergone 12 years of clinical validation, has helped bring tens of thousands of healthy babies into the world, and has received certifications from multiple regions including the US FDA, European CE, and Australian TGA. Its technological barriers are clear, placing it in the top tier of the industry.
First, R&D risk is eliminated. By acquiring a mature product directly, Basecare has bypassed years of R&D investment and the uncertainty of potential R&D failure, enabling rapid product implementation and market launch, thus shortening the performance delivery cycle. This is a significant advantage for a target with clear expectations for performance improvement.
Second, an existing market foundation is in place. This product was previously authorized for sale by COOK in China, where it has captured 40% of the domestic market share. This means its market acceptance has been fully validated. After Basecare takes over, it can rapidly achieve market penetration without needing to re-cultivate the market, converting the existing market share into its own revenue, providing clear support for performance increment.
Third, demand side exhibits rigidity. Clinical data shows that this product can increase the rate of high-quality embryos by 46%, effectively improving the pregnancy success rate for older couples and reducing miscarriage rates. With domestic demand for fertility at an advanced age continuously rising and the volume of assisted reproductive treatments growing steadily, hospitals' rigid demand for high-quality consumables provides stable support for the product's subsequent sales, mitigating concerns about demand falling short of expectations.
II. Industry Analysis: Domestic Substitution + Policy Dividends, Sector Prosperity Continues to Rise
"Sector prosperity" serves as a core dimension for assessing a target's long-term value. The assisted reproduction sector in which Basecare operates is currently at the intersection of two major trends: "accelerated domestic substitution + release of policy dividends." The industry's prosperity is continuously rising, forming the core logic for its long-term investment value.
Previously, the domestic embryo culture medium market was long dominated by overseas giants, characterized not only by relatively high prices but also by supply shortage risks due to international supply chain fluctuations. The need for domestic substitution was urgent. The approval of Basecare's related product marks a significant breakthrough in the domestic substitution of high-end embryo culture media, precisely aligning with the broader trend of domestic substitution for medical devices in China, offering inherent policy advantages.
Firstly, policy dividends are continuously increasing. The national level persistently promotes fertility support policies. The gradual inclusion of assisted reproductive technology in medical insurance and the implementation of childcare subsidies directly boost the volume of assisted reproductive treatments, thereby driving demand for related consumables. This provides clear policy support for Basecare's product sales, ensuring a stable demand side for the industry.
Secondly, product competitiveness holds differentiated advantages. Compared to imported products, Basecare's domestically produced version offers higher cost-effectiveness. It can also be paired with the company's already approved supporting equipment, providing hospitals with a one-stop solution. This synergistic advantage can effectively enhance market competitiveness, accelerate the import substitution process, and seize market share from overseas giants.
Thirdly, revenue growth has clear expectations. Referring to the product's previous sales performance in China, if fully rolled out subsequently, it is expected to bring stable revenue increments to Basecare, becoming a new engine for the company's performance growth. This also serves as a core catalytic factor for short-term performance improvement.
III. Company Transformation: Full-Chain Layout Realized, Valuation Reassessment Holds Imaginative Potential
From the perspective of the company's development logic, this product approval serves as a key marker of Basecare's strategic transformation implementation. Previously, the company mainly focused on the preimplantation genetic testing (PGT) business, with a relatively single business scope and weaker risk resistance capability. Through the acquisition of relevant international brands, the company has successfully opened up the full-chain layout of "Testing + Equipment + Consumables," completing its transformation from a single IVD (in vitro diagnostics) enterprise to a full-chain IVF service provider, further improving its business matrix.
Post-transformation, Basecare can not only provide testing services but also sell products such as embryo culture media and culture equipment. The business synergy is significant, effectively enhancing value per client. More crucially, currently, few domestic enterprises have achieved a full-chain layout in assisted reproduction. This differentiated positioning helps the company break free from the growth bottleneck of a single business and enhance its long-term competitiveness.
From a valuation logic perspective, an improved business structure helps reduce the operational risks associated with a single business, enhances the stability of the company's profitability, and creates room for a re-evaluation of the company's valuation. For long-term investors, this transformation towards a full-chain layout serves as a core manifestation of the company's long-term development potential and a key support for subsequent valuation reassessment.
IV. Investment Analysis and Risk Warning: View Positive News Rationally, Beware of Volatility Risks
Based on the above analysis, the investment value assessment for Basecare Medical (02170.HK) is as follows: In the short term, the product approval serves as a clear positive catalyst. Coupled with the product's high maturity and solid market foundation, short-term performance growth has a certain degree of certainty, which is expected to drive a valuation repair for the stock price. In the long term, the realization of the full-chain layout, the advancement of domestic substitution, coupled with rising industry prosperity, suggest the company has certain growth potential.
However, investors are reminded to remain rational. The realization of positive news does not necessarily equate to a stock price increase. Relevant risks still need to be monitored, with three key points to focus on:
1. Performance Realization Risk: Post-approval, the progress of market promotion and customer expansion may fall short of expectations, leading to slower-than-expected revenue growth. This serves as the most critical risk to watch in the short term.
2. Market Competition Risk: Overseas giants will not easily give up market share and may compete through price reductions, product upgrades, etc. If Basecare cannot achieve rapid market penetration, it may impact performance growth.
3. Policy and Market Volatility Risk: The assisted reproduction industry is significantly influenced by policy changes. Adjustments to related policies could affect industry demand. Concurrently, the Hong Kong stock market itself is highly volatile; short-term positive news might be digested by market sentiment. Investors need to implement risk control measures.
Comprehensive Assessment: The approval of Basecare's Gems one-step embryo culture medium represents a significant milestone in the company's business development and a key step in its strategic transformation and domestic substitution layout. It aligns with the industry development trend and possesses certain investment merit. For small and medium investors, Basecare can be considered a key watchlist target in the assisted reproduction sector. In the long run, with the popularization of assisted reproduction inclusion in medical insurance and the increase in industry concentration, Basecare, as a leading enterprise in the upstream of the industrial chain, its technological barriers and the value of its full industrial chain layout are likely to be repriced by the market. The company's valuation system stands at a critical juncture of reshaping. The current stock price does not fully reflect its potential in high-barrier technology platforms and future performance conversion, indicating significant room for value reassessment and appreciation.
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

Basecare Medical (HK.2170) Receives NMPA Approval for Core Product Geri Medium, Unlocking New Opportunities in Assisted Reproduction

Basecare Medical (HK.2170) Receives NMPA Approval for Core Product Geri Medium, Unlocking New Opportunities in Assisted Reproduction
Basecare Medical (HK.2170) Receives NMPA Approval for Core Product Geri Medium, Unlocking New Opportunities in Assisted Reproduction

Basecare Medical (HK.2170) Receives NMPA Approval for Core Product Geri Medium, Unlocking New Opportunities in Assisted Reproduction