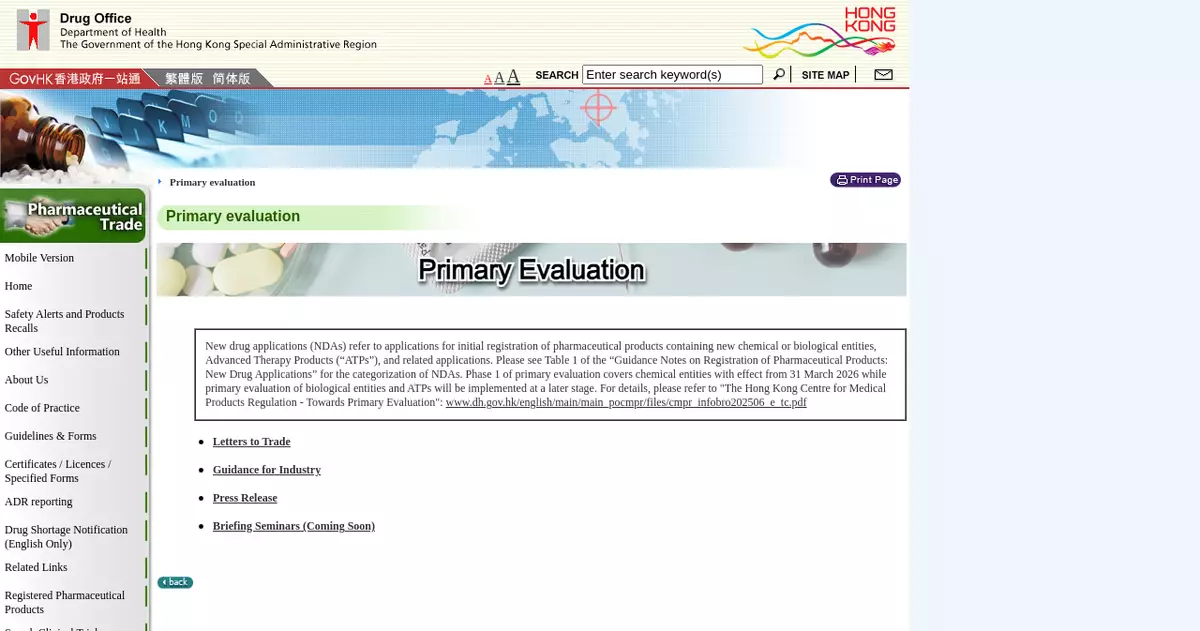
"Primary evaluation" begins phased implementation and applications of new drugs for registration accepted under initial phase
The Department of Health (DH) today (March 31) began the phased implementation of "primary evaluation" for new drug registration. The initial phase covers applications for the registration of drug products containing locally registered chemical entities with extended applications (e.g. new indications, new strengths, new posology, new dosage forms, etc). Applications are accepted from today. The DH has sent letters to notify pharmaceutical associations and stakeholders (including holders and applicants of certificates of drug registration), and will organise online briefing seminars next month to explain the relevant details.
As mentioned in "The Chief Executive's 2025 Policy Address" that the Hong Kong Special Administrative Region (HKSAR) Government would establish the Hong Kong Centre for Medical Products Regulation (CMPR) in 2026 and begin the phased implementation of the "primary evaluation" mechanism for new drug registration in the same year. The HKSAR Government aims to achieve full coverage of all pharmaceutical products by 2030, which will enable Hong Kong to independently assess and approve the safety and efficacy of new drugs based on clinical data, with a view to expediting the introduction of innovative medical products, thereby achieving the goal of making good drugs and medical devices available for use in Hong Kong for the benefit of patients.
To pave the way for "primary evaluation", the Government first implemented the "1+" drug approval mechanism on November 1, 2023, to facilitate the registration of new drugs in Hong Kong for treating life-threatening or severely debilitating diseases. The mechanism was extended on November 1 the following year to cover all new drugs, including all new chemical or biological entities, new indications, vaccines and advanced therapy products. New drugs that are supported by local clinical data and recognised by local experts can be applied for registration in Hong Kong if the applicant submits evidence of approval from the drug regulatory authority of one non-local reference place (instead of two in the past). Since the launch of the "1+" mechanism, 19 new drugs have been approved under this mechanism. Seven of them have been listed in the Hospital Authority Drug Formulary. This brings new treatment options for patients and attracts more drug research and development (R&D) as well as clinical trials to be conducted in Hong Kong.
"Primary evaluation" is a new and comprehensive regulatory process to approve applications for registration of new drugs which involves the independent assessment of primary data of all pre-clinical trials (i.e. animal testing), clinical studies, pharmacovigilance, manufacturing and quality control, etc in order to fully ensure their safety, efficacy and quality before and after the drugs are placed in the market (i.e. throughout the product life cycle). Registering new drugs under the "primary evaluation" mechanism facilitates immediate approval without relying and waiting for approval from non-local drug regulatory authorities, which further speeds up the introduction of new drugs and clinical applications. This will not only help speed up patients' access to breakthrough treatments but also boost medical R&D, testing and related industries in Hong Kong. The Office for Introducing Innovative Drugs and Medical Devices under the Hospital Authority will, after ascertaining the needs and benefits of innovative drug treatments for local patients, proactively liaise with manufacturers of innovative drugs and medical devices, and utilise the "1+" mechanism for registration to bring in innovative drugs and medical devices that are beneficial and cost-effective for patients.
For details of applications for the registration and the briefing seminars, please visit the DH's dedicated website.
Source: AI-found images