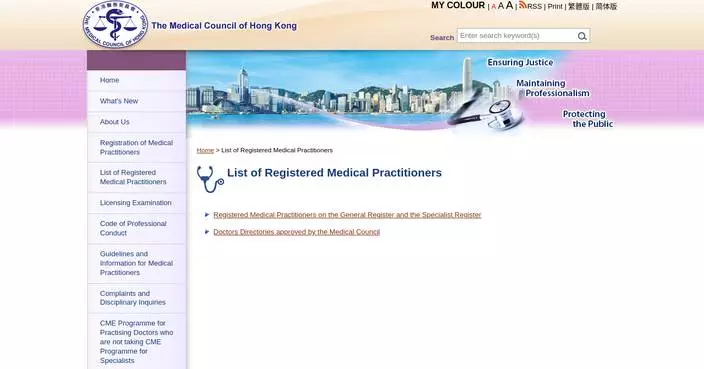
Hong Kong Customs seizes suspected illegally imported live birds
Hong Kong Customs today (May 8) seized 242 illegally imported live birds, with an estimated market value of about $48,400 at the Lok Ma Chau Spur Line Control Point.
Customs officers today intercepted an incoming 67-year-old male passenger at the arrival hall of the said control point. Upon examination, 242 live birds contained in bird cages were seized from his personal baggage, and he was subsequently arrested. The case was handed over to the Agriculture, Fisheries and Conservation Department for follow-up investigation.
Under the Public Health (Animals and Birds) Regulations, it is an offence to import any bird unless it is accompanied by a valid health certificate. The maximum penalty upon conviction is a fine of $25,000.
Members of the public may report any suspected activities of illegal import of live birds to Customs' 24-hour hotline 182 8080 or its dedicated crime-reporting email account (crimereport@customs.gov.hk) or online form (eform.cefs.gov.hk/form/ced002).

Hong Kong Customs today (May 8) detected a suspected case of illegal importing of live birds at the Lok Ma Chau Spur Line Control Point. 242 live birds with an estimated market value of about $48,400 were seized. Photo shows the suspected illegally imported live birds seized. Source: HKSAR Government Press Releases
DH follows up on medical incident reported by Tuen Mun Hospital
In response to Tuen Mun Hospital (TMH)'s announcement of a sentinel event, the Department of Health (DH) today (May 15) said that the DH is investigating the incident and following up with the local responsible person (LRP) of the medical device concerned.
On May 13, the DH received a notification from the Hospital Authority (HA) indicating that air bubbles had been detected in the coronary artery of a 75-year-old male patient during a Percutaneous Coronary Intervention (PCI) performed earlier at TMH. According to the preliminary information provided by the HA, it is suspected that the incident is related to the pressure monitoring set used during the procedure.
As the pressure monitoring set in question is listed under the DH's Medical Device Administrative Control System (MDACS), upon receiving the notification, the DH immediately took the following follow-up actions:
 Contacted the LRP of the medical device concerned to ascertain the details and requested the LRP to conduct an investigation and submit an investigation report;
As a precautionary measure, the LRP has been instructed to immediately suspend sales of the affected batch and to contact all users in Hong Kong who are using medical devices from the same batch to advise them to temporarily stop using the affected product. To date, the DH has not received any adverse event reporting regarding this medical device from other users; and
Issued a Special Alert on the DH's Medical Device Division website and notified stakeholders, urging users who possess the affected product to temporarily suspend their use and contact the LRP for follow up.
Contacted the LRP of the medical device concerned to ascertain the details and requested the LRP to conduct an investigation and submit an investigation report;
As a precautionary measure, the LRP has been instructed to immediately suspend sales of the affected batch and to contact all users in Hong Kong who are using medical devices from the same batch to advise them to temporarily stop using the affected product. To date, the DH has not received any adverse event reporting regarding this medical device from other users; and
Issued a Special Alert on the DH's Medical Device Division website and notified stakeholders, urging users who possess the affected product to temporarily suspend their use and contact the LRP for follow up.
DH's investigation is ongoing. The DH will continue to maintain close communication with the LRP, closely monitor the situation and take follow-up actions based on the investigation report.
The DH introduced the voluntary Medical Device Administrative Control System (MDACS) in 2004, under which a listing system for medical devices and traders as well as a post-market monitoring system for the products are put in place. All listed medical devices meet the safety, quality and performance requirements. Currently, over 9 000 medical devices are listed under the MDACS. Over the past three years, the DH has not received any safety alerts or adverse event reports involving the pressure monitoring set concerned. In response to the latest international regulatory trends, the Government is formulating the statutory regulatory requirements and will consult various stakeholders as appropriate within this year before submitting the legislative proposal to the Legislative Council. The implementation of a statutory registration system for medical devices aligns with the policy directions set out by the Chief Executive in the 2023, 2024 and 2025 Policy Addresses to accelerate the implementation of reforms to the evaluation, approval and registration mechanism for medical products. The HKSAR Government plans to introduce a bill in the second half of this year to achieve the goal of establishing the Hong Kong Centre for Medical Products Regulation (CMPR) under the DH by the end of 2026. Upon its establishment, the CMPR will systematically consolidate the regulatory functions for Western and Chinese medicines as well as medical devices. By leveraging existing legislation governing Western and Chinese medicines and the proposed statutory regulatory framework for medical devices, the CMPR will comprehensively enhance the regulatory and approval mechanism for medical products, thereby supporting Hong Kong's development as an international Health and Medical Innovation Hub.

Source: AI-found images