|
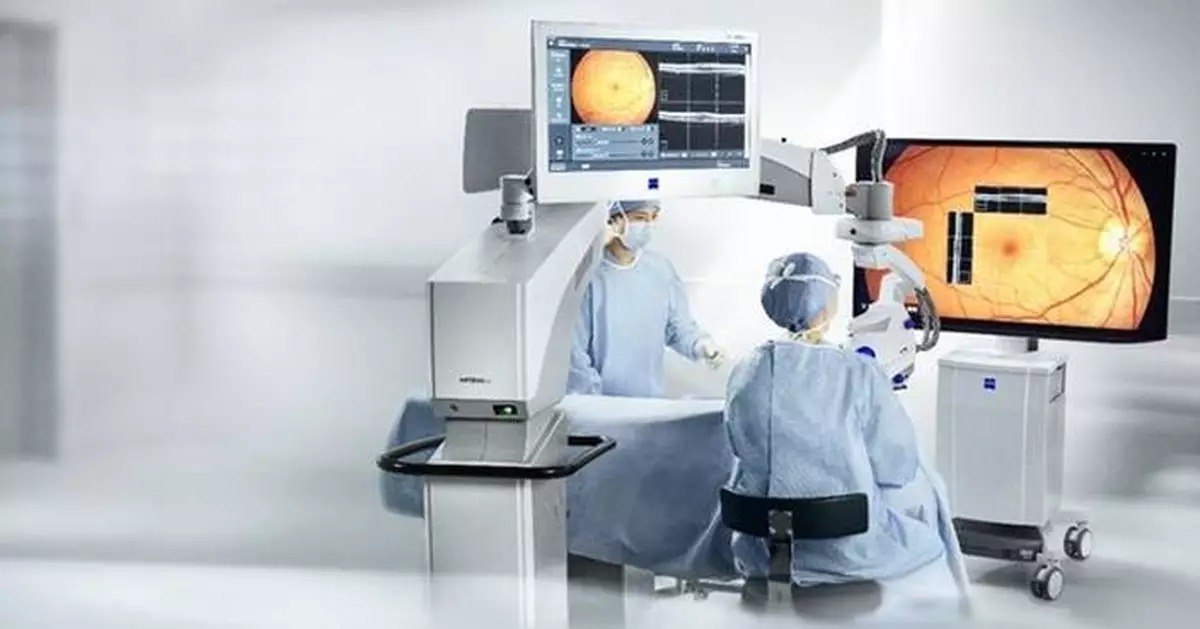
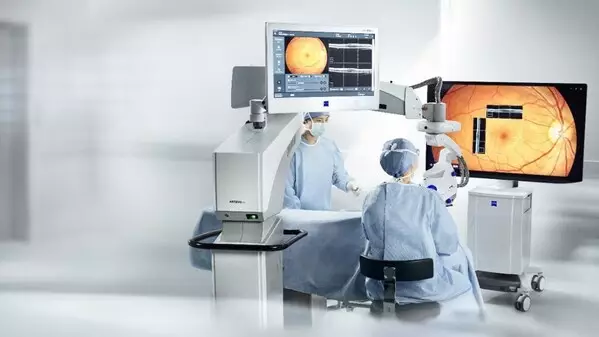
- Elevates digital visualization for surgeons - ZEISS ARTEVO 850 surgical microscope provides ophthalmic surgeons in Mexico with advanced 3D visualization and end-to-end workflow integration.
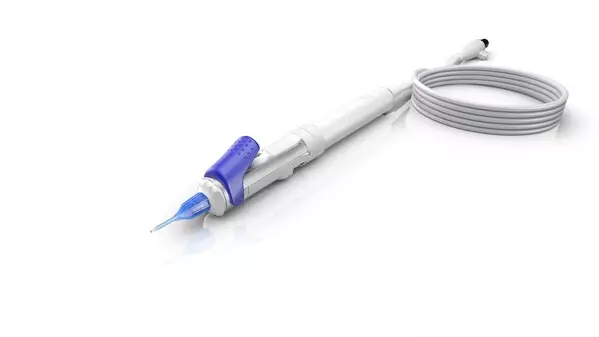
- Minimizes risk and increases O.R. efficiency for cataract surgery - ZEISS MICOR 700 first hand-held lens removal device with ultrasound-free operation reinvents lens extraction by providing a sustainable solution with a low initial investment to help cataract surgeons expand their intraocular working space.
JENA, Germany and MEXICO CITY, May 13, 2026 /PRNewswire/ -- ZEISS Medical Technology announced today that Mexico's Comisión Federal para la Protección contra Riesgos Sanitários (COFEPRIS) approved the ARTEVO® 850 ophthalmic microscope from ZEISS and the MICOR® 700 from ZEISS. Both technologies offer full compatibility with the latest generation of ZEISS's cataract workflow. The ZEISS ARTEVO 850 and the ZEISS MICOR 700 will be showcased as part of ZEISS's ophthalmic workflows in the ZEISS booth #A1 at the XXXVIII Mexican Congress of Ophthalmology (SMO) in Monterrey, Mexico, from May 30 – June 3, 2026.
"ZEISS continues to expand its portfolio around the world, this time adding new surgical solutions in Mexico. We're investing in regional markets to empower local surgeons with the latest in visual and O.R. enhancements, helping surgeons provide their patients with the best eye care possible," says PT Cheong, Head of Global Sales for Ophthalmology, ZEISS Medical Technology.
"The ZEISS MICOR 700 and ZEISS ARTEVO 850 are true game changers in cataract surgery, elevating visualization and the workflow in the operating room. The introduction of these solutions into the Mexican market helps enable surgeons to operate with convenient control and efficiency," says Frank Seitzinger, Head of Business Sector Surgery Anterior Segment, ZEISS Medical Technology.
The ZEISS ARTEVO 850 3D heads-up ophthalmic microscope sets the pace in digital visualization with true color imaging and increased depth of field by nearly 60 percent.1 The surgical microscope also offers functional integration of devices and visualization technology to streamline clinical procedures. Digital integration into the ZEISS Ophthalmic Workflows allows for seamless data transfer. Additionally, the ZEISS ARTEVO 850 with CALLISTO eye® features a redesigned intuitive user interface that centralizes all controls on a single touchscreen.
The MICOR® 700 from ZEISS reinvents lens extraction with the first hand-held lens removal device with ultrasound-free operation, using the ZEISS NULEX (non-ultrasonic lens extraction) procedure to help surgeons broaden their intraocular working space, minimizing risk to surrounding eye structures and increasing operating room efficiency. The tip of the ZEISS MICOR 700 is designed to be gentle with the surrounding tissue. Its blunt tip design with rounded edges is designed to minimize the risk of tissue damage. ZEISS MICOR 700 also features a disposable "plug-and-play" system ensuring convenience, efficiency, and a minimal O.R. footprint.
For more information about ZEISS ophthalmic workflow solutions in Mexico, visit here.
1 Data on file. Compared to ZEISS ARTEVO 800.
Not all products, services or offers are approved or offered in every market and approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development. The statements of the healthcare professionals reflect only their personal opinions and experiences and do not necessarily reflect the opinion of any institution that they are affiliated with. The healthcare professionals alone are responsible for the content of their experience reported and any potential resulting infringements. Carl Zeiss Meditec AG and its affiliates to not have clinical evidence supporting the opinions and statements of the health care professionals nor accept any responsibility or liability of the healthcare professionals' content. The healthcare professionals have a contractual or other financial relationship with Carl Zeiss Meditec AG and its affiliates and have received financial support.
Contact for investors
Sebastian Frericks
Director Investor Relations
Carl Zeiss Meditec AG
Phone: +49 3641 220 116
Mail: investors.med@zeiss.com
Contact for the press
Frank Smith
Head of Global Communications
Ophthalmology, Carl Zeiss Meditec Inc.
Phone: +49 3641 220 331
Mail: press.med@zeiss.com
www.zeiss.com/newsroom
Brief Profile
Carl Zeiss Meditec AG (ISIN: DE0005313704), which is listed on the MDAX and TecDAX of the German stock exchange, is one of the world's leading medical technology companies. The Company supplies innovative technologies and application-oriented solutions designed to help doctors improve the quality of life of their patients. The Company offers complete solutions, including implants and consumables, to diagnose and treat eye diseases. The Company creates innovative visualization solutions in the field of microsurgery. With 5,730 employees worldwide, the Group generated revenue of €2,066.1m in fiscal year 2023/24 (to 30 September).
The Group's head office is located in Jena, Germany, and it has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the USA, Japan, Spain and France. The Center for Application and Research (CARIn) in Bangalore, India and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, strengthen the Company's presence in these rapidly developing economies. Around 39 percent of Carl Zeiss Meditec AG's shares are in free float. Approx. 59 percent are held by Carl Zeiss AG, one of the world's leading groups in the optical and optoelectronic industries.
For further information visit: www.zeiss.com/med
- Elevates digital visualization for surgeons - ZEISS ARTEVO 850 surgical microscope provides ophthalmic surgeons in Mexico with advanced 3D visualization and end-to-end workflow integration.
- Minimizes risk and increases O.R. efficiency for cataract surgery - ZEISS MICOR 700 first hand-held lens removal device with ultrasound-free operation reinvents lens extraction by providing a sustainable solution with a low initial investment to help cataract surgeons expand their intraocular working space.
JENA, Germany and MEXICO CITY, May 13, 2026 /PRNewswire/ -- ZEISS Medical Technology announced today that Mexico's Comisión Federal para la Protección contra Riesgos Sanitários (COFEPRIS) approved the ARTEVO® 850 ophthalmic microscope from ZEISS and the MICOR® 700 from ZEISS. Both technologies offer full compatibility with the latest generation of ZEISS's cataract workflow. The ZEISS ARTEVO 850 and the ZEISS MICOR 700 will be showcased as part of ZEISS's ophthalmic workflows in the ZEISS booth #A1 at the XXXVIII Mexican Congress of Ophthalmology (SMO) in Monterrey, Mexico, from May 30 – June 3, 2026.
"ZEISS continues to expand its portfolio around the world, this time adding new surgical solutions in Mexico. We're investing in regional markets to empower local surgeons with the latest in visual and O.R. enhancements, helping surgeons provide their patients with the best eye care possible," says PT Cheong, Head of Global Sales for Ophthalmology, ZEISS Medical Technology.
"The ZEISS MICOR 700 and ZEISS ARTEVO 850 are true game changers in cataract surgery, elevating visualization and the workflow in the operating room. The introduction of these solutions into the Mexican market helps enable surgeons to operate with convenient control and efficiency," says Frank Seitzinger, Head of Business Sector Surgery Anterior Segment, ZEISS Medical Technology.
The ZEISS ARTEVO 850 3D heads-up ophthalmic microscope sets the pace in digital visualization with true color imaging and increased depth of field by nearly 60 percent.1 The surgical microscope also offers functional integration of devices and visualization technology to streamline clinical procedures. Digital integration into the ZEISS Ophthalmic Workflows allows for seamless data transfer. Additionally, the ZEISS ARTEVO 850 with CALLISTO eye® features a redesigned intuitive user interface that centralizes all controls on a single touchscreen.
The MICOR® 700 from ZEISS reinvents lens extraction with the first hand-held lens removal device with ultrasound-free operation, using the ZEISS NULEX (non-ultrasonic lens extraction) procedure to help surgeons broaden their intraocular working space, minimizing risk to surrounding eye structures and increasing operating room efficiency. The tip of the ZEISS MICOR 700 is designed to be gentle with the surrounding tissue. Its blunt tip design with rounded edges is designed to minimize the risk of tissue damage. ZEISS MICOR 700 also features a disposable "plug-and-play" system ensuring convenience, efficiency, and a minimal O.R. footprint.
For more information about ZEISS ophthalmic workflow solutions in Mexico, visit here.
1 Data on file. Compared to ZEISS ARTEVO 800.
Not all products, services or offers are approved or offered in every market and approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development. The statements of the healthcare professionals reflect only their personal opinions and experiences and do not necessarily reflect the opinion of any institution that they are affiliated with. The healthcare professionals alone are responsible for the content of their experience reported and any potential resulting infringements. Carl Zeiss Meditec AG and its affiliates to not have clinical evidence supporting the opinions and statements of the health care professionals nor accept any responsibility or liability of the healthcare professionals' content. The healthcare professionals have a contractual or other financial relationship with Carl Zeiss Meditec AG and its affiliates and have received financial support.
Contact for investors
Sebastian Frericks
Director Investor Relations
Carl Zeiss Meditec AG
Phone: +49 3641 220 116
Mail: investors.med@zeiss.com
Contact for the press
Frank Smith
Head of Global Communications
Ophthalmology, Carl Zeiss Meditec Inc.
Phone: +49 3641 220 331
Mail: press.med@zeiss.com
www.zeiss.com/newsroom
Brief Profile
Carl Zeiss Meditec AG (ISIN: DE0005313704), which is listed on the MDAX and TecDAX of the German stock exchange, is one of the world's leading medical technology companies. The Company supplies innovative technologies and application-oriented solutions designed to help doctors improve the quality of life of their patients. The Company offers complete solutions, including implants and consumables, to diagnose and treat eye diseases. The Company creates innovative visualization solutions in the field of microsurgery. With 5,730 employees worldwide, the Group generated revenue of €2,066.1m in fiscal year 2023/24 (to 30 September).
The Group's head office is located in Jena, Germany, and it has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the USA, Japan, Spain and France. The Center for Application and Research (CARIn) in Bangalore, India and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, strengthen the Company's presence in these rapidly developing economies. Around 39 percent of Carl Zeiss Meditec AG's shares are in free float. Approx. 59 percent are held by Carl Zeiss AG, one of the world's leading groups in the optical and optoelectronic industries.
For further information visit: www.zeiss.com/med
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
ZEISS expands ophthalmic surgical workflows in Mexico with COFEPRIS approval for ARTEVO 850 and MICOR 700
ZEISS expands ophthalmic surgical workflows in Mexico with COFEPRIS approval for ARTEVO 850 and MICOR 700
TAIPEI, May 13, 2026 /PRNewswire/ -- The S&P Global Sustainability Yearbook Awards Ceremony was held in Taipei on May 13. TECO Electric & Machinery Co., Ltd. (TWSE: 1504) was named to the 2026 edition of The Sustainability Yearbook by global index and ratings agency S&P Global for the sixth time, in recognition of its long-standing commitment to sustainable management.
TECO has also been selected for the Dow Jones Best-in-Class Indices (DJBICI), underscoring its continued progress across environmental, social, and governance dimensions. TECO Chairman Morris Li stated that sustainable development is a core element of TECO's long-term competitiveness. Guided by its focus on electrification, intelligence, and green energy, TECO will further advance ESG governance, operational carbon reduction, and low-carbon innovation in line with international sustainability standards, strengthening its resilience amid the global transition toward net zero.
In recent years, TECO Group has actively embedded sustainability into its global operations. TECO has set a target to reduce operational emissions by 50% over a ten-year period by 2030 and, by 2025, had achieved a reduction of more than 40%. TECO has also continued to install solar power generation facilities at its own sites, with global installed capacity exceeding 17 MW in the same year, steadily advancing toward its 2030 goal of having renewable energy account for 30% of the Group's total electricity consumption.
To translate carbon reduction commitments into concrete management mechanisms, TECO has introduced an internal carbon pricing mechanism of NT$1,600 per metric ton of CO2e and established an internal carbon fund mechanism to support investments in energy management, renewable energy, low-carbon innovation, and capability building. Through this mechanism, three low-carbon proposals were launched last year, with expected annual carbon reductions approaching 1,000 metric tons of CO2e. TECO has also implemented an energy management program and publicly disclosed energy consumption information to strengthen energy performance management, enhance operational transparency, and continuously improve energy efficiency.
Beyond reducing emissions from its own operations, TECO has extended its sustainability influence to supply chain and industry partners. For two consecutive years, TECO has participated in the "Net-Zero Transformation Promotion Program for the Manufacturing Sector" initiated by the Industrial Development Administration, Ministry of Economic Affairs. Working with more than 20 corporate partners, TECO has formed a "1+N Carbon Management Demonstration Team" to help supply chain partners strengthen carbon management and low-carbon transformation capabilities through carbon inventory assessments and in-depth energy-saving guidance.
In corporate governance, TECO's Board of Directors oversees climate-related issues and sustainability strategies. TECO has also linked concrete KPIs, including carbon reduction results, carbon intensity, and green supply chain performance, to senior executives' performance evaluations, establishing a dual mechanism of "governance plus incentives" to ensure that sustainability goals are effectively integrated into organizational management and operational performance.
As TECO marks its 70th anniversary this year, TECO will take this milestone as an important step toward its next stage of growth and transformation, continuing to deepen its "B (Business) 2B (Business) 2S (Sustainability)" philosophy and working with global customers and suppliers to move together toward a net-zero future.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

TECO Earns Global ESG Recognition with S&P Global Sustainability Yearbook and Dow Jones Best-in-Class Indices Inclusion