NEW YORK, Jan. 2, 2025 /PRNewswire/ -- PieX AI, a global leader in multimodal AI hardware for smart health, is set to proudly launch the world's first pendant featuring personalized, on-device AI at CES 2025. This innovative product aims to revolutionize mental health management by seamlessly integrating technology with emotional well-being.
A Focus on Mental Health
PieX's mission is to enhance users' mental health through advanced applications such as AI life coaching, guided breathing exercises, and comprehensive health monitoring. The PieX pendant is a holistic tool that applies physiological and psychological to support emotional well-being. By empowering users to recognize and embrace their feelings, PieX promotes users to build a positive mindset, ultimately enhancing their mental energy and well-being.
Proprietary Sensing and AI Technology
The PieX pendant is built upon our proprietary Sensing Technology and foundation models featuring both Personalized AI and On-device AI. The sensing technology provides 24/7 accurate emotion tracking results to build a personalized emotion database for each user, making the AI know users better. Moreover, the AI running on users' own phones ensures that users' emotional data and auto-journaled information are processed and stored locally, eliminating the need for cloud storage. This feature provides an ultimate solution to enhance user privacy and data security in the LLM era.
- Sensing Tech: The pendant's sensing capability not only accurately measures indicators of stress, such as the wearer's HRV (heart rate variability), but also precisely assesses the user's emotional state through their speech tone during conversation. By combining these two aspects, it provides users with a comprehensive emotional tracking insight. Moreover, the pendant's sensing ability also offers users the capacity to perceive their environment and events, correlating emotional states with daily occurrences, thus helping users better cope with various emotional changes.
- AI Tech: Leveraging the exceptional sensing capabilities of the pendant, our device will provide a personalized AI that understands the user best. It can automatically offer options for meditation and breathing exercises based on the user's emotional changes and preferences, enabling users to take a more proactive approach to their mental health. Additionally, all emotional sensing and data processing analysis occur solely on the user's personal device, with no need for cloud uploads. Even large language models run locally, ensuring that sensitive information never leaves the user's devices.
PieX AI at CES: Booth #63001-02,Level 1,Hall G, Venetian Expo.
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

PieX AI: Launching the World's First Personalized, On-device AI Pendant at CES 2025
|
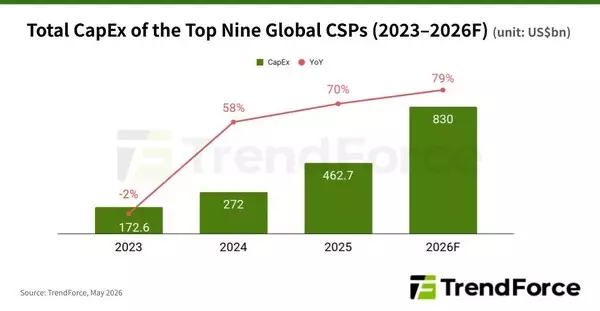
NEW YORK, May 6, 2026 /PRNewswire/ -- TrendForce's latest findings on the AI industry highlight that several major North American CSPs have recently raised their 2026 capital expenditure (CapEx) guidance in response to strong AI demand. As a result, TrendForce has revised its forecast for the combined CapEx of the world's top nine CSPs—Google, AWS, Meta, Microsoft, Oracle, ByteDance, Tencent, Alibaba, and Baidu—up to approximately US$830 billion in 2026, with the annual growth rate raised from 61% to 79%.
Taking a look at the four major U.S. CSPs, Microsoft has increased its CapEx outlook to $190 billion, implying approximately 130% YoY growth. Around roughly $25 billion is attributable to rising component costs. Similarly, Google has raised its guidance from $175–185 billion to $180–190 billion, with growth exceeding 100%. Meta has revised its CapEx range upward from $115–135 billion to $125–145 billion, representing approximately 85% YoY growth. Lastly, AWS is expected to exceed $230 billion in CapEx this year, with growth of over 50% driven by demand for AI cloud services.
TrendForce notes that the pace of CapEx expansion among North American CSPs exceeds the global average, underscoring that AI infrastructure has become a core long-term strategic priority. Investment is increasingly concentrated in the deployment of high-performance GPU clusters, in-house ASIC development, and next-generation data centers designed to support high-power-density computing.
The sharp rise in CapEx also signals sustained momentum in data center construction, led primarily by AWS, Microsoft, Google, Meta, and Oracle. As of the end of 2025, these five North American CSPs had deployed 800–900 data centers globally, with AWS accounting for the lion's share.
Among Chinese CSPs, Alibaba and ByteDance are the main drivers of expansion—though their strategies diverge. Alibaba is focusing on localized nodes and sovereign cloud offerings through Alibaba Cloud to penetrate emerging markets. Since announcing expansion plans in 2025, the company has established new regions in Brazil, France, and the Netherlands, enlarging its global footprint to 29 regions and 94 availability zones.
In contrast, ByteDance is aggressively expanding overseas through TikTok, with operations established across eight countries, including the U.S., Brazil, and Ireland, and major investments in Europe, Thailand, and Malaysia. This makes it the most geographically aggressive Chinese CSP.
TrendForce further notes that sustained AI demand will continue to drive global data center growth, with total installed power capacity expected to reach approximately 155 GW in 2026 (~+29% YoY). AI servers are also projected to surpass general-purpose servers in total electricity consumption in 2026 due to significantly higher power consumption per unit.
Further jumps in power consumption are expected in 2027–2028 as platforms such as GB300/Rubin and ASIC-based AI servers enter mass production. This trend will, in turn, support growth in key components such as HVDC power systems and liquid cooling systems.
For more information on TrendForce's semiconductor reports and market data, please visit the Report Page, or Email (SR_MI@trendforce.com) the Sales Department.
For more on the latest technology industry news and trends, please visit News.
About TrendForce
TrendForce is a global leader in technology industry analysis and consulting services. With deep expertise spanning foundry, DRAM, HBM, NAND Flash, AI servers, robotics, near-eye displays, display panels, LEDs, MLCC, and green energy, it also offers in-depth research into key market drivers such as AI, automotive technologies, 5G/6G communications, LEO satellites, and the IoT.
Backed by a team of top industry professionals, TrendForce has been at the forefront of global market research for over 25 years. More than 60% of its clients are among the world's top 500 companies.
TrendForce's global footprint includes Taipei, Shenzhen, Silicon Valley, New York, and Tokyo. With timely and strategic industry analysis, TrendForce delivers the critical information that empowers businesses to make smarter, faster decisions.
CONTACT: pengchen@trendforce.com
NEW YORK, May 6, 2026 /PRNewswire/ -- TrendForce's latest findings on the AI industry highlight that several major North American CSPs have recently raised their 2026 capital expenditure (CapEx) guidance in response to strong AI demand. As a result, TrendForce has revised its forecast for the combined CapEx of the world's top nine CSPs—Google, AWS, Meta, Microsoft, Oracle, ByteDance, Tencent, Alibaba, and Baidu—up to approximately US$830 billion in 2026, with the annual growth rate raised from 61% to 79%.
Taking a look at the four major U.S. CSPs, Microsoft has increased its CapEx outlook to $190 billion, implying approximately 130% YoY growth. Around roughly $25 billion is attributable to rising component costs. Similarly, Google has raised its guidance from $175–185 billion to $180–190 billion, with growth exceeding 100%. Meta has revised its CapEx range upward from $115–135 billion to $125–145 billion, representing approximately 85% YoY growth. Lastly, AWS is expected to exceed $230 billion in CapEx this year, with growth of over 50% driven by demand for AI cloud services.
TrendForce notes that the pace of CapEx expansion among North American CSPs exceeds the global average, underscoring that AI infrastructure has become a core long-term strategic priority. Investment is increasingly concentrated in the deployment of high-performance GPU clusters, in-house ASIC development, and next-generation data centers designed to support high-power-density computing.
The sharp rise in CapEx also signals sustained momentum in data center construction, led primarily by AWS, Microsoft, Google, Meta, and Oracle. As of the end of 2025, these five North American CSPs had deployed 800–900 data centers globally, with AWS accounting for the lion's share.
Among Chinese CSPs, Alibaba and ByteDance are the main drivers of expansion—though their strategies diverge. Alibaba is focusing on localized nodes and sovereign cloud offerings through Alibaba Cloud to penetrate emerging markets. Since announcing expansion plans in 2025, the company has established new regions in Brazil, France, and the Netherlands, enlarging its global footprint to 29 regions and 94 availability zones.
In contrast, ByteDance is aggressively expanding overseas through TikTok, with operations established across eight countries, including the U.S., Brazil, and Ireland, and major investments in Europe, Thailand, and Malaysia. This makes it the most geographically aggressive Chinese CSP.
TrendForce further notes that sustained AI demand will continue to drive global data center growth, with total installed power capacity expected to reach approximately 155 GW in 2026 (~+29% YoY). AI servers are also projected to surpass general-purpose servers in total electricity consumption in 2026 due to significantly higher power consumption per unit.
Further jumps in power consumption are expected in 2027–2028 as platforms such as GB300/Rubin and ASIC-based AI servers enter mass production. This trend will, in turn, support growth in key components such as HVDC power systems and liquid cooling systems.
For more information on TrendForce's semiconductor reports and market data, please visit the Report Page, or Email (SR_MI@trendforce.com) the Sales Department.
For more on the latest technology industry news and trends, please visit News.
About TrendForce
TrendForce is a global leader in technology industry analysis and consulting services. With deep expertise spanning foundry, DRAM, HBM, NAND Flash, AI servers, robotics, near-eye displays, display panels, LEDs, MLCC, and green energy, it also offers in-depth research into key market drivers such as AI, automotive technologies, 5G/6G communications, LEO satellites, and the IoT.
Backed by a team of top industry professionals, TrendForce has been at the forefront of global market research for over 25 years. More than 60% of its clients are among the world's top 500 companies.
TrendForce's global footprint includes Taipei, Shenzhen, Silicon Valley, New York, and Tokyo. With timely and strategic industry analysis, TrendForce delivers the critical information that empowers businesses to make smarter, faster decisions.
CONTACT: pengchen@trendforce.com
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
North American AI Data Center Expansion Drives 2026 CapEx of Top Nine CSPs to US$830 Billion, Says TrendForce











