|
The excimer laser complements the ZEISS VISUMAX 800 with ZEISS SMILE pro, extending ZEISS' LVC market leadership with treatment for myopia, hyperopia, and mixed astigmatism.
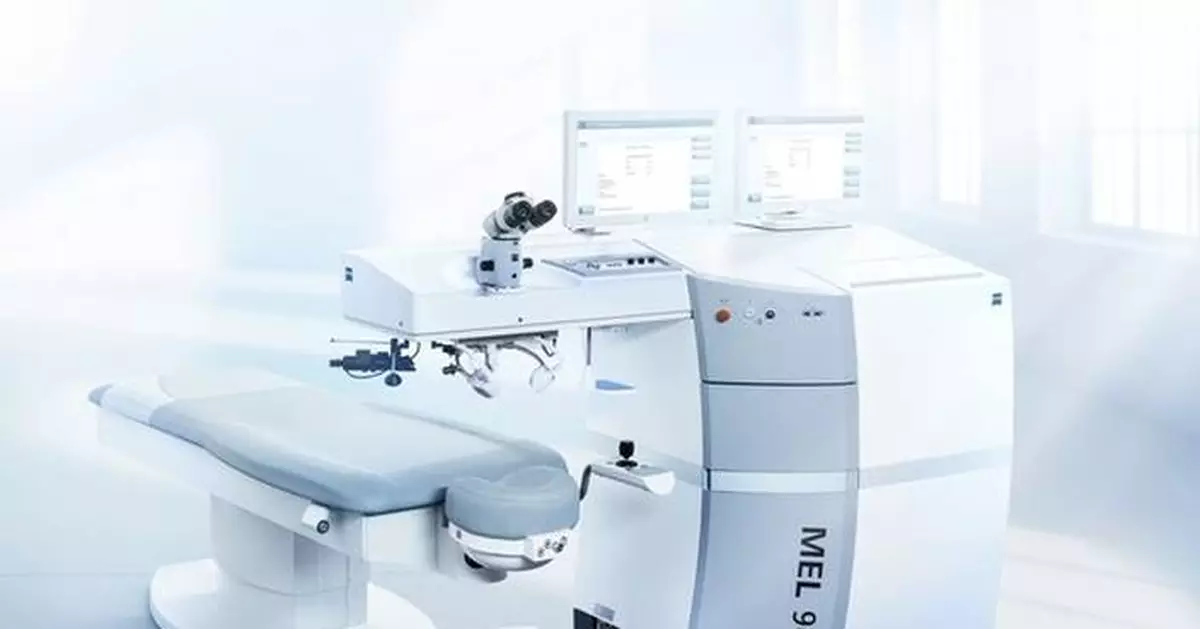
DUBLIN, Calif. and JENA, Germany, Jan. 13, 2025 /PRNewswire/ -- ZEISS Medical Technology announced today that the MEL® 90 received approval from the U.S. Food and Drug Administration (FDA), giving the excimer laser technology simultaneous approval for all three major indications, including myopia, hyperopia and mixed astigmatism (a condition where both hyperopic and myopic correction is required). The ZEISS MEL 90 fully integrates into the Corneal Refractive Workflow to offer U.S. surgeons a fast, reliable, and streamlined surgery experience, complementing the VISUMAX® 800 with SMILE® pro from ZEISS to help further broaden a surgeon's refractive business with improved patient attraction and positive clinical outcomes.
"The increasing global adoption of laser vision correction reflects the advancements and positive impact the technology continues to have on the quality of life for patients," says Magnus Reibenspiess, Head of Strategic Business Unit Ophthalmology at ZEISS Medical Technology. "With the integration of the ZEISS MEL 90, surgeons can confidently care for their patients with greater workflow efficiency and performance with enhanced outcomes."
"ZEISS continues to break new ground as a leader in the LVC market, reflecting our ongoing commitment to and support of U.S. surgeons and patients with the latest refractive innovation that helps set practices apart and provides more options to more patients," says Andrew Chang, Head of Global Sales for ZEISS Medical Technology. "With the availability of the ZEISS MEL 90 in the U.S. market, surgeons can now offer an integrated workflow of refractive technology to help reach new patients and provide excellent outcomes for them."
"The FDA approval of the ZEISS MEL 90 excimer laser is a game changer for refractive surgery in the U.S. This advanced technology, with its Triple-A ablation algorithm and fast ablation speed, sets a new benchmark for precision, safety, and efficiency. I am particularly impressed by the system's ability to simplify treatment planning while delivering highly predictable and tissue-conserving results. The integration of the ZEISS MEL 90 with the ZEISS VISUMAX 800 femtosecond laser opens new doors for streamlined workflows and optimal patient outcomes. This approval marks an exciting chapter for surgeons and patients alike," says Dr. John Doane, Refractive Surgeon, Discover Vision Centers, Kansas City, MO.
Reliable Outcomes: The ZEISS MEL 90 offers Triple-A (Advanced Ablation Algorithm), which is a single ablation profile for a wide range of sphero-cylindrical (SCA) corrections that simplifies treatment planning. Triple-A offers a high degree of accuracy, reproducibility and predictability, as well as advantages such as tissue-saving ablation. Its preinstalled functions also offer surgeons optimal support for achieving excellent treatment results and gentle treatments of standard, higher and lower levels of ametropia and astigmatism.
Fast Ablation Speed: The ZEISS MEL 90 offers a truly customized power package. When performing LASIK for myopia at 500 Hz, the ZEISS MEL 90 can intra-operatively ablate 1 diopter in as little as 1.3 seconds.1 Intuitive system guidance and speedy treatment planning allow for additional time savings. The ZEISS MEL 90 excimer laser technology is equipped with an active eye tracker which delivers an excellent response time, providing a high level of treatment safety with very stable results.
Easy to Use: New capabilities of the ZEISS MEL 90 excimer laser greatly simplify the interactions between the surgeon, the assisting staff and the system technology. The excimer laser can be configured to precisely match the surgeon and the OR team's needs. The system's simple, intuitive graphic user interface supports fast treatment procedures, and the touch screen can be flexibly positioned wherever it is needed to enable the surgeon to have good ergonomic sitting posture throughout an operation. A second, optional touch screen can be configured to optimally complement the treatment routine of the OR team. An HD video port, network printer connection, and PDF export capabilities offer additional workflow support.
With the ZEISS MEL 90, the company expands its offerings for U.S. refractive clinics and helps support the improvement of clinical outcomes and patient satisfaction through onboarding, training, service support and ZEISS Practice Development Consulting.
The ZEISS MEL 90 excimer laser is currently available in the U.S. For more information, visit www.zeiss.com/us/mel-90
1LASIK myopia, optical zone 6 mm.
Not all products, services or offers are approved or offered in every market and approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development. The statements of the healthcare professionals reflect only their personal opinions and experiences and do not necessarily reflect the opinion of any institution that they are affiliated with. The healthcare professionals alone are responsible for the content of their experience reported and any potential resulting infringements. Carl Zeiss Meditec AG and its affiliates to not have clinical evidence supporting the opinions and statements of the health care professionals nor accept any responsibility or liability of the healthcare professionals' content. The healthcare professionals have a contractual or other financial relationship with Carl Zeiss Meditec AG and its affiliates and have received financial support.
Brief profile
Carl Zeiss Meditec AG (ISIN: DE0005313704), which is listed on the MDAX and TecDAX of the German stock exchange, is one of the world's leading medical technology companies. The Company supplies innovative technologies and application-oriented solutions designed to help doctors improve the quality of life of their patients. The Company offers complete solutions, including implants and consumables, to diagnose and treat eye diseases. The Company creates innovative visualization solutions in the field of microsurgery. With 5,730 employees worldwide, the Group generated revenue of €2,066.1m in fiscal year 2023/24 (to 30 September).
The Group's head office is located in Jena, Germany, and it has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the USA, Japan, Spain and France. The Center for Application and Research (CARIn) in Bangalore, India and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, strengthen the Company's presence in these rapidly developing economies. Around 39 percent of Carl Zeiss Meditec AG's shares are in free float. Approx. 59 percent are held by Carl Zeiss AG, one of the world's leading groups in the optical and optoelectronic industries.
For more information visit our website at www.zeiss.com/med
The excimer laser complements the ZEISS VISUMAX 800 with ZEISS SMILE pro, extending ZEISS' LVC market leadership with treatment for myopia, hyperopia, and mixed astigmatism.
DUBLIN, Calif. and JENA, Germany, Jan. 13, 2025 /PRNewswire/ -- ZEISS Medical Technology announced today that the MEL® 90 received approval from the U.S. Food and Drug Administration (FDA), giving the excimer laser technology simultaneous approval for all three major indications, including myopia, hyperopia and mixed astigmatism (a condition where both hyperopic and myopic correction is required). The ZEISS MEL 90 fully integrates into the Corneal Refractive Workflow to offer U.S. surgeons a fast, reliable, and streamlined surgery experience, complementing the VISUMAX® 800 with SMILE® pro from ZEISS to help further broaden a surgeon's refractive business with improved patient attraction and positive clinical outcomes.
"The increasing global adoption of laser vision correction reflects the advancements and positive impact the technology continues to have on the quality of life for patients," says Magnus Reibenspiess, Head of Strategic Business Unit Ophthalmology at ZEISS Medical Technology. "With the integration of the ZEISS MEL 90, surgeons can confidently care for their patients with greater workflow efficiency and performance with enhanced outcomes."
"ZEISS continues to break new ground as a leader in the LVC market, reflecting our ongoing commitment to and support of U.S. surgeons and patients with the latest refractive innovation that helps set practices apart and provides more options to more patients," says Andrew Chang, Head of Global Sales for ZEISS Medical Technology. "With the availability of the ZEISS MEL 90 in the U.S. market, surgeons can now offer an integrated workflow of refractive technology to help reach new patients and provide excellent outcomes for them."
"The FDA approval of the ZEISS MEL 90 excimer laser is a game changer for refractive surgery in the U.S. This advanced technology, with its Triple-A ablation algorithm and fast ablation speed, sets a new benchmark for precision, safety, and efficiency. I am particularly impressed by the system's ability to simplify treatment planning while delivering highly predictable and tissue-conserving results. The integration of the ZEISS MEL 90 with the ZEISS VISUMAX 800 femtosecond laser opens new doors for streamlined workflows and optimal patient outcomes. This approval marks an exciting chapter for surgeons and patients alike," says Dr. John Doane, Refractive Surgeon, Discover Vision Centers, Kansas City, MO.
Reliable Outcomes: The ZEISS MEL 90 offers Triple-A (Advanced Ablation Algorithm), which is a single ablation profile for a wide range of sphero-cylindrical (SCA) corrections that simplifies treatment planning. Triple-A offers a high degree of accuracy, reproducibility and predictability, as well as advantages such as tissue-saving ablation. Its preinstalled functions also offer surgeons optimal support for achieving excellent treatment results and gentle treatments of standard, higher and lower levels of ametropia and astigmatism.
Fast Ablation Speed: The ZEISS MEL 90 offers a truly customized power package. When performing LASIK for myopia at 500 Hz, the ZEISS MEL 90 can intra-operatively ablate 1 diopter in as little as 1.3 seconds.1 Intuitive system guidance and speedy treatment planning allow for additional time savings. The ZEISS MEL 90 excimer laser technology is equipped with an active eye tracker which delivers an excellent response time, providing a high level of treatment safety with very stable results.
Easy to Use: New capabilities of the ZEISS MEL 90 excimer laser greatly simplify the interactions between the surgeon, the assisting staff and the system technology. The excimer laser can be configured to precisely match the surgeon and the OR team's needs. The system's simple, intuitive graphic user interface supports fast treatment procedures, and the touch screen can be flexibly positioned wherever it is needed to enable the surgeon to have good ergonomic sitting posture throughout an operation. A second, optional touch screen can be configured to optimally complement the treatment routine of the OR team. An HD video port, network printer connection, and PDF export capabilities offer additional workflow support.
With the ZEISS MEL 90, the company expands its offerings for U.S. refractive clinics and helps support the improvement of clinical outcomes and patient satisfaction through onboarding, training, service support and ZEISS Practice Development Consulting.
The ZEISS MEL 90 excimer laser is currently available in the U.S. For more information, visit www.zeiss.com/us/mel-90
1LASIK myopia, optical zone 6 mm.
Not all products, services or offers are approved or offered in every market and approved labeling and instructions may vary from one country to another. For country-specific product information, see the appropriate country website. Product specifications are subject to change in design and scope of delivery as a result of ongoing technical development. The statements of the healthcare professionals reflect only their personal opinions and experiences and do not necessarily reflect the opinion of any institution that they are affiliated with. The healthcare professionals alone are responsible for the content of their experience reported and any potential resulting infringements. Carl Zeiss Meditec AG and its affiliates to not have clinical evidence supporting the opinions and statements of the health care professionals nor accept any responsibility or liability of the healthcare professionals' content. The healthcare professionals have a contractual or other financial relationship with Carl Zeiss Meditec AG and its affiliates and have received financial support.
Brief profile
Carl Zeiss Meditec AG (ISIN: DE0005313704), which is listed on the MDAX and TecDAX of the German stock exchange, is one of the world's leading medical technology companies. The Company supplies innovative technologies and application-oriented solutions designed to help doctors improve the quality of life of their patients. The Company offers complete solutions, including implants and consumables, to diagnose and treat eye diseases. The Company creates innovative visualization solutions in the field of microsurgery. With 5,730 employees worldwide, the Group generated revenue of €2,066.1m in fiscal year 2023/24 (to 30 September).
The Group's head office is located in Jena, Germany, and it has subsidiaries in Germany and abroad; more than 50 percent of its employees are based in the USA, Japan, Spain and France. The Center for Application and Research (CARIn) in Bangalore, India and the Carl Zeiss Innovations Center for Research and Development in Shanghai, China, strengthen the Company's presence in these rapidly developing economies. Around 39 percent of Carl Zeiss Meditec AG's shares are in free float. Approx. 59 percent are held by Carl Zeiss AG, one of the world's leading groups in the optical and optoelectronic industries.
For more information visit our website at www.zeiss.com/med
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

ZEISS MEL 90 excimer laser receives U.S. FDA approval; completes Corneal Refractive Workflow
|
The First Time a Game in the Legendary Franchise Comes to Consoles
BELLEVUE, Wash., June 6, 2026 /PRNewswire/ -- Critically-acclaimed game development studio ArenaNet® tonight announced Guild Wars 3™, the third entry in the legendary and beloved Guild Wars® MMORPG franchise and their first new game since 2012. The game will release globally on PC, Steam, and PlayStation 5, marking the first time a Guild Wars game will appear on home consoles. The first beta test is scheduled for fall of 2027.
The official debut trailer features the first in-engine gameplay footage and can be viewed HERE. The first concept art and other assets are hosted at the Guild Wars 3 official press kit.
"Guild Wars 3 is a new era not just for ArenaNet and Guild Wars, but also for MMORPGs as a whole," said Colin Johanson, Guild Wars 3 Game Director and ArenaNet Studio Head. "The MMORPG genre has millions of players worldwide eager for new, modern experiences, and the opportunity to harness that excitement is immense. Our approach with Guild Wars 3 is to push MMORPGs forward, to create an online game world that feels believable, rewarding, responsive, and innovative while at the same time respecting players' time and providing a positive, supportive space to build community and enjoy new stories in our universe."
Taking place over a thousand years before the events of the original game, Guild Wars 3 is set in the Tyrian region of Orr, a vast wilderness frontier imbued with the world's magic. Nature entities with strong connections to the land called Vael spirits embody the vitality of this lush, verdant landscape. Various guilds are engaged in conflict over how to protect or exploit the bounty beyond civilization's borders. These multitudinous spirits will vary in size and influence within the ecosystems around them, with the most notable of these being the Seeker. As seen in the trailer, every player's Seeker will serve as a connection to the spirits of Orr and a mount that will carry them on their journeys through the game's open world.
Players will assume the role of a Vaelwarden, a member of a guild of adventurers committed to preserving and protecting both the spirits of the wild and the land of Orr itself. Through the deep character customization and skill-building gameplay the Guild Wars franchise is known for, players will seek adventure and confront danger across this sumptuous, vibrant online world, growing their relationships with the spirits of the wild, the inhabitants of Orr, and other Vaelwarden players.
A combat system designed from the outset to feel great on both a controller and keyboard rewards positioning by emphasizing the joy of movement and momentum. As players seamlessly transition between various movement modes, they can harness their speed and turn it into bigger damage and impact when fighting their foes.
More information about pricing, beta tests, release date, and gameplay features will be revealed later in 2026 and into 2027. Guild Wars 3 will be available directly from the developers at GuildWars3.com, and can be wishlisted on Steam and the PlayStation Store beginning today, with more information available at the game's official website.
About the Guild Wars Franchise
Guild Wars is the award-winning, visually stunning MMORPG franchise that originally launched in 2005 on PC. Boasting a timeless art-brought-to-life aesthetic, the epic grandeur of massive role-playing environments, fast-paced action combat, large-scale dynamic events that players tackle together in the open world, team-based competitive player-versus-player battles, and a sprawling fantasy storyline, the series is often proclaimed as having the friendliest player community in gaming. Guild Wars was relaunched as Guild Wars Reforged in 2025 and expanded to mobile devices in 2026. Guild Wars 2® launched in 2012 on PC and quickly earned accolades across the industry, including Time Magazine's 2012 Game of the Year. Unlike most competitors, the franchise has always been playable with no subscription fee required, welcoming over 29 million players across its history.
About ArenaNet
ArenaNet is the developer of the best-selling Guild Wars Reforged and the groundbreaking Guild Wars 2 massively multiplayer online PC role-playing games. The studio's mission is to create innovative online worlds, cultivate a vibrant and engaged global community of players, and to incorporate handcrafted artistry into every aspect of their games. ArenaNet formed in 2000, with press and fans consistently calling Guild Wars and Guild Wars 2 two of the best MMOs and RPGs of all time. Based in Bellevue, WA, ArenaNet is a wholly-owned subsidiary of Korean gaming giant NC.
The First Time a Game in the Legendary Franchise Comes to Consoles
BELLEVUE, Wash., June 6, 2026 /PRNewswire/ -- Critically-acclaimed game development studio ArenaNet® tonight announced Guild Wars 3™, the third entry in the legendary and beloved Guild Wars® MMORPG franchise and their first new game since 2012. The game will release globally on PC, Steam, and PlayStation 5, marking the first time a Guild Wars game will appear on home consoles. The first beta test is scheduled for fall of 2027.
The official debut trailer features the first in-engine gameplay footage and can be viewed HERE. The first concept art and other assets are hosted at the Guild Wars 3 official press kit.
"Guild Wars 3 is a new era not just for ArenaNet and Guild Wars, but also for MMORPGs as a whole," said Colin Johanson, Guild Wars 3 Game Director and ArenaNet Studio Head. "The MMORPG genre has millions of players worldwide eager for new, modern experiences, and the opportunity to harness that excitement is immense. Our approach with Guild Wars 3 is to push MMORPGs forward, to create an online game world that feels believable, rewarding, responsive, and innovative while at the same time respecting players' time and providing a positive, supportive space to build community and enjoy new stories in our universe."
Taking place over a thousand years before the events of the original game, Guild Wars 3 is set in the Tyrian region of Orr, a vast wilderness frontier imbued with the world's magic. Nature entities with strong connections to the land called Vael spirits embody the vitality of this lush, verdant landscape. Various guilds are engaged in conflict over how to protect or exploit the bounty beyond civilization's borders. These multitudinous spirits will vary in size and influence within the ecosystems around them, with the most notable of these being the Seeker. As seen in the trailer, every player's Seeker will serve as a connection to the spirits of Orr and a mount that will carry them on their journeys through the game's open world.
Players will assume the role of a Vaelwarden, a member of a guild of adventurers committed to preserving and protecting both the spirits of the wild and the land of Orr itself. Through the deep character customization and skill-building gameplay the Guild Wars franchise is known for, players will seek adventure and confront danger across this sumptuous, vibrant online world, growing their relationships with the spirits of the wild, the inhabitants of Orr, and other Vaelwarden players.
A combat system designed from the outset to feel great on both a controller and keyboard rewards positioning by emphasizing the joy of movement and momentum. As players seamlessly transition between various movement modes, they can harness their speed and turn it into bigger damage and impact when fighting their foes.
More information about pricing, beta tests, release date, and gameplay features will be revealed later in 2026 and into 2027. Guild Wars 3 will be available directly from the developers at GuildWars3.com, and can be wishlisted on Steam and the PlayStation Store beginning today, with more information available at the game's official website.
About the Guild Wars Franchise
Guild Wars is the award-winning, visually stunning MMORPG franchise that originally launched in 2005 on PC. Boasting a timeless art-brought-to-life aesthetic, the epic grandeur of massive role-playing environments, fast-paced action combat, large-scale dynamic events that players tackle together in the open world, team-based competitive player-versus-player battles, and a sprawling fantasy storyline, the series is often proclaimed as having the friendliest player community in gaming. Guild Wars was relaunched as Guild Wars Reforged in 2025 and expanded to mobile devices in 2026. Guild Wars 2® launched in 2012 on PC and quickly earned accolades across the industry, including Time Magazine's 2012 Game of the Year. Unlike most competitors, the franchise has always been playable with no subscription fee required, welcoming over 29 million players across its history.
About ArenaNet
ArenaNet is the developer of the best-selling Guild Wars Reforged and the groundbreaking Guild Wars 2 massively multiplayer online PC role-playing games. The studio's mission is to create innovative online worlds, cultivate a vibrant and engaged global community of players, and to incorporate handcrafted artistry into every aspect of their games. ArenaNet formed in 2000, with press and fans consistently calling Guild Wars and Guild Wars 2 two of the best MMOs and RPGs of all time. Based in Bellevue, WA, ArenaNet is a wholly-owned subsidiary of Korean gaming giant NC.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

Guild Wars 3, Modern Evolution of the MMORPG, Announced at Summer Game Fest for PC and PlayStation 5