- Enhancing Clinical Efficiency with AI Sleep Analysis: Cutting Both Cost and Time
- Joint Clinical Studies with CROs: SOMNUM™ AI Sleep Scoring Drives Higher Efficiency
- FDA-cleared AI solution shortens clinical trial timelines while ensuring data reliability
- Expanded global partnerships with CROs enable a large-scale clinical data analysis platform
BOSTON, Nov. 24, 2025 /PRNewswire/ -- HoneyNaps, an AI-driven sleep-medicine technology company, announced that its AI-based sleep disorder diagnostic support software, SOMNUM™, which recently received clearance from the U.S. Food and Drug Administration (FDA), is significantly reducing clinical trial timelines and costs for pharmaceutical and medical device companies through enhanced data-analysis efficiency.
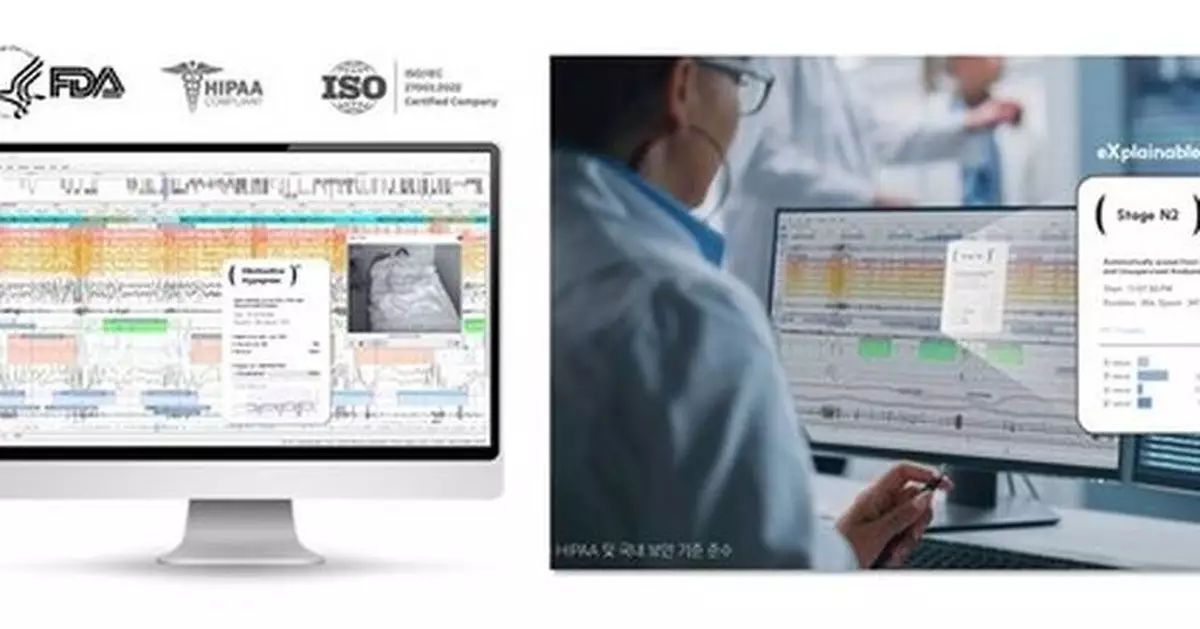
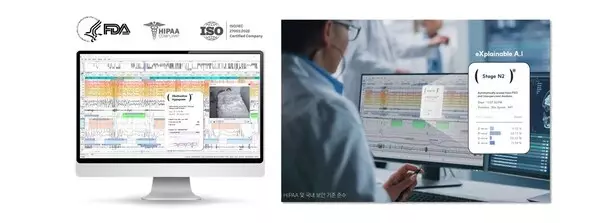
SOMNUM™ is a medical AI software solution designed to automatically analyze polysomnography (PSG) data. The system identifies sleep stages and key events—including apnea, hypopnea, and arousals—using advanced artificial intelligence–based PSG scoring.
Following its FDA 510(k) clearance last year, SOMNUM™ has been formally recognized as a validated analytical tool for sleep-related clinical trials and medical device evaluations. This clearance underscores the safety and reliability of AI-based sleep-analysis technology as acknowledged by global regulatory authorities.
Traditionally, PSG scoring required specialists to manually review and classify data for more than three to four hours per case, resulting in high labor and time costs. SOMNUM™ automates this process, shortening scoring time to just minutes, while clinicians review and verify the results to maintain diagnostic quality.
By integrating SOMNUM™, pharmaceutical companies and medical device manufacturers have been able to accelerate clinical trial pipelines, increase data-processing speed by more than 70%, and reduce overall study timelines by approximately 50%. Scoring labor costs and quality-control (QC) expenses have also dropped by 80–90%.
HoneyNaps is expanding its global clinical-market presence with SOMNUM™ at the core of its business strategy. The company is currently in discussions with U.S. pharmaceutical firms regarding the application of SOMNUM™ in sleep-related clinical studies and has established strategic partnerships with multiple CROs. Through these collaborations, HoneyNaps provides a large-scale biosignal-analysis platform essential for sleep-medicine and neurological clinical trials—delivering both accelerated pipelines and enhanced data accuracy.
The company is also offering an AI-based re-scoring (verification scoring) service for already-scored PSG datasets. This new business model provides an objective check on the accuracy of existing scoring results. CROs leverage this re-scoring solution to secure data quality while shortening trial timelines and strengthening the reliability of data submitted to regulatory bodies. Beyond simple verification, the service supports clients in advancing future projects, proprietary algorithms, and technology development—ultimately enhancing R&D competitiveness.
SOMNUM™ goes beyond conventional AI analysis by incorporating explainable AI (XAI) technology, enabling clinicians to review the rationale behind the AI's scoring decisions. This ensures transparency and verifiability of trial data, meeting the stringent quality standards required by global regulatory agencies.
HoneyNaps is currently collaborating with major tertiary hospitals and sleep centers in Korea, as well as university hospitals and clinical institutions across the United States and Europe, to expand its SOMNUM™-based AI analysis platform. The technology is being used not only in clinical trials for sleep and wake-promoting therapeutics, but also in evaluating the effectiveness of CPAP devices, sleep sensors, wearable devices, and a range of sleep-technology products.
Taekyoung (Sean) Ha, PhD, President of HoneyNaps USA, stated, "SOMNUM's PSG AI scoring technology is fundamentally transforming the traditionally time-consuming manual scoring process that has long been a bottleneck in sleep-related clinical trials. Backed by FDA clearance, SOMNUM™ is now delivering tangible improvements in research efficiency and cost reduction for global pharmaceutical and medical device companies."
For further information, please contact:
HoneyNaps USA, Inc.
Christine Kwon / Managing Director
Email: sleep@honeynaps.com
Address: #517, SPACES, 361 Newbury Street, Boston, MA, 02115
BOSTON, Nov. 24, 2025 /PRNewswire/ -- HoneyNaps, an AI-driven sleep-medicine technology company, announced that its AI-based sleep disorder diagnostic support software, SOMNUM™, which recently received clearance from the U.S. Food and Drug Administration (FDA), is significantly reducing clinical trial timelines and costs for pharmaceutical and medical device companies through enhanced data-analysis efficiency.
SOMNUM™ is a medical AI software solution designed to automatically analyze polysomnography (PSG) data. The system identifies sleep stages and key events—including apnea, hypopnea, and arousals—using advanced artificial intelligence–based PSG scoring.
Following its FDA 510(k) clearance last year, SOMNUM™ has been formally recognized as a validated analytical tool for sleep-related clinical trials and medical device evaluations. This clearance underscores the safety and reliability of AI-based sleep-analysis technology as acknowledged by global regulatory authorities.
Traditionally, PSG scoring required specialists to manually review and classify data for more than three to four hours per case, resulting in high labor and time costs. SOMNUM™ automates this process, shortening scoring time to just minutes, while clinicians review and verify the results to maintain diagnostic quality.
By integrating SOMNUM™, pharmaceutical companies and medical device manufacturers have been able to accelerate clinical trial pipelines, increase data-processing speed by more than 70%, and reduce overall study timelines by approximately 50%. Scoring labor costs and quality-control (QC) expenses have also dropped by 80–90%.
HoneyNaps is expanding its global clinical-market presence with SOMNUM™ at the core of its business strategy. The company is currently in discussions with U.S. pharmaceutical firms regarding the application of SOMNUM™ in sleep-related clinical studies and has established strategic partnerships with multiple CROs. Through these collaborations, HoneyNaps provides a large-scale biosignal-analysis platform essential for sleep-medicine and neurological clinical trials—delivering both accelerated pipelines and enhanced data accuracy.
The company is also offering an AI-based re-scoring (verification scoring) service for already-scored PSG datasets. This new business model provides an objective check on the accuracy of existing scoring results. CROs leverage this re-scoring solution to secure data quality while shortening trial timelines and strengthening the reliability of data submitted to regulatory bodies. Beyond simple verification, the service supports clients in advancing future projects, proprietary algorithms, and technology development—ultimately enhancing R&D competitiveness.
SOMNUM™ goes beyond conventional AI analysis by incorporating explainable AI (XAI) technology, enabling clinicians to review the rationale behind the AI's scoring decisions. This ensures transparency and verifiability of trial data, meeting the stringent quality standards required by global regulatory agencies.
HoneyNaps is currently collaborating with major tertiary hospitals and sleep centers in Korea, as well as university hospitals and clinical institutions across the United States and Europe, to expand its SOMNUM™-based AI analysis platform. The technology is being used not only in clinical trials for sleep and wake-promoting therapeutics, but also in evaluating the effectiveness of CPAP devices, sleep sensors, wearable devices, and a range of sleep-technology products.
Taekyoung (Sean) Ha, PhD, President of HoneyNaps USA, stated, "SOMNUM's PSG AI scoring technology is fundamentally transforming the traditionally time-consuming manual scoring process that has long been a bottleneck in sleep-related clinical trials. Backed by FDA clearance, SOMNUM™ is now delivering tangible improvements in research efficiency and cost reduction for global pharmaceutical and medical device companies."
For further information, please contact:
HoneyNaps USA, Inc.
Christine Kwon / Managing Director
Email: sleep@honeynaps.com
Address: #517, SPACES, 361 Newbury Street, Boston, MA, 02115
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **
HoneyNaps Accelerates Clinical Trial Pipelines with FDA-Cleared AI "SOMNUM"