TAIPEI, Dec. 9, 2025 /PRNewswire/ --
Data Center AI Chip Shipments Continue to Grow
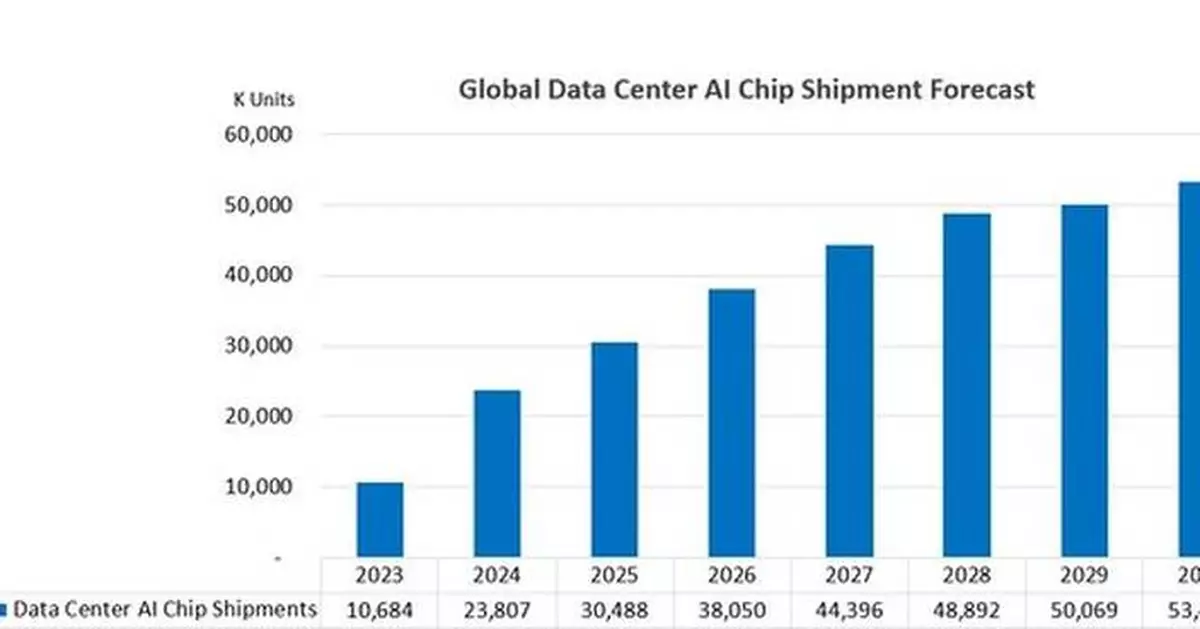
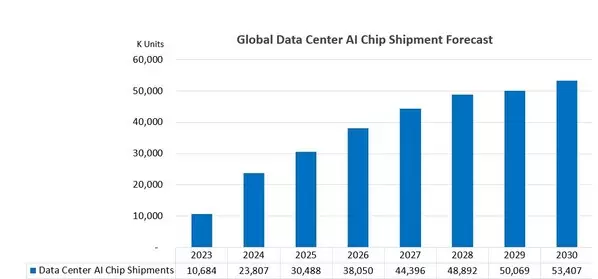
According to the latest report published by DIGITIMES Asia, global data center AI chip shipments are projected to grow from 30.5 million units in 2024 to 53.4 million units in 2030. This data center AI chip category includes high-end and mid-range GPUs, application-specific AI chips (such as Google's TPUs), AI server CPUs, and networking/interconnect-related chips (e.g., Switch ASICs/rack-scale-up Interconnect Chips/DPUs & NICs).
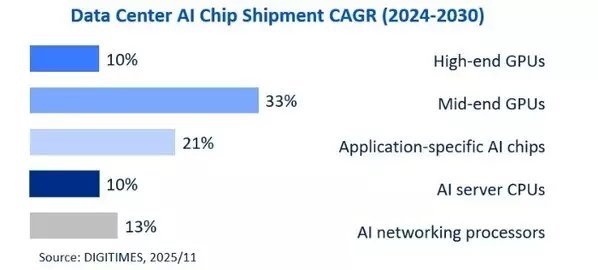
In terms of growth rate, the fastest-growing segments by shipment volume include application-specific AI chips, mid-range GPUs that use GDDR DRAM (such as Nvidia CPX GPUs), and interconnect-related chips, such as NVSwitch ICs. Among these five chip categories, the shipment volume growth rates for high-end GPUs and AI server CPUs are relatively lower over the 2024–2030 period, with a Compound Annual Growth Rate (CAGR) of only about 10%. However, when considering the revenue from high-end packaging and end-market sales, the growth rate will be much higher than the shipment growth rate. The main reasons for this include the increased content value per chip and the adoption of advanced packaging.
For example, AMD recently forecast that the global server CPU revenue Total Addressable Market (TAM) CAGR will reach 18% between 2025 and 2030 (implying that the Average Selling Price CAGR for server CPUs could exceed 7% due to the jump in core count and the adoption of more advanced processes and packaging). For GPUs, despite relatively moderate growth in shipments, wafer foundry and packaging revenue CAGRs are both expected to exceed 40% in 2024-2030, driven by the increasing number of GPU and I/O dies per chip.
Data Center AI Chip Packaging Revenue Remains Focused on GPUs
Recently, application-specific AI chips like Google's TPU and AWS's Trainium have garnered significant attention for their tailored power-efficiency characteristics, sparking speculation that they might replace GPUs. According to the latest "Global Data Center AI Chip Packaging Market Forecast 2024-2030" report published by DIGITIMES Asia, the overall revenue share of data center AI chip packaging will remain GPU-centric throughout the 2024–2030 forecast period. It is estimated that by 2030, packaging revenue for data center GPUs will remain over 40% higher than that of application-specific AI chips.
While AI server CPUs and AI networking-related chips have high shipment volumes, the high-end packaging market will still be overwhelmingly dominated by GPUs and application-specific AI chips.
Drivers and Inhibitors for Advanced Packaging of Data Center AI Chips
DIGITIMES Asia forecasts that the global market size for advanced packaging of data center AI chips will grow from US$5.6 billion in 2024 to US$53.1 billion in 2030, representing a CAGR of over 40%. This aligns closely with AMD's recent Financial Analyst Day estimate that the global Data Center TAM CAGR will exceed 40% over the next five years.
Here is a summary of the main drivers and inhibitors for the global market for advanced packaging of data center AI chips:
Key Drivers:
- The AI wave and the compute power arms race among tech giants.
- Advanced packaging driving system-level scaling to extend Moore's Law.
- Geopolitical competition between the U.S. and China, and the rise of Sovereign AI initiatives.
Key Inhibitors:
- The re-evaluation of return on investment (ROI) relative to massive computing power investments.
- The impact of cheaper and more efficient new technologies.
For more details on AI Chip, and the future outlook for major AI chip packaging technologies in data centers, check out: AI Chip packaging https://www.digitimes.com/reports/ai/2025_ai_chip_packaging
** The press release content is from PR Newswire. Bastille Post is not involved in its creation. **

Strong Momentum expected for Data Center AI Chip Packaging in 2025-2030
Strong Momentum expected for Data Center AI Chip Packaging in 2025-2030
Strong Momentum expected for Data Center AI Chip Packaging in 2025-2030
|
SOMERSET, N.J., March 3, 2026 /PRNewswire/ -- Harvest Integrated Research Organization (HiRO), a global contract research organization specializing in strategic planning for clinical development and cross‑border clinical trial solutions and services, concluded a successful presence at the 44th Annual J.P. Morgan Healthcare Conference in San Francisco.
HiRO's Founder and CEO, Dr. Karen Chu, was a featured speaker at RESI JPM, where she moderated a panel convening investors and strategics at the forefront of the cell and gene therapy revolution. Panelists Robert Balfour of ALSA Ventures and Bettina Ernst of BERNINA BioInvest highlighted the current momentum in cell and gene therapies, discussed how they assess technical and commercial risk, identified partnership models that accelerate progress, and outlined where capital is flowing in this rapidly advancing field.
Dr. Chu also led a workshop titled "Leveraging Asia: How to Navigate Asian VC Investment Mandates." The session opened with the 2026 industry outlook and key trends, then highlighted priority therapeutic areas. It explained the NewCo model and the criteria Asian venture capital firms use to evaluate global biotech assets, their co‑investment preferences, and expectations for commercialization. The workshop featured Asia‑based investors Dr. Alva Chen, Managing Director and Head of Therapeutics, VMS Group; Jayson Lee, Partner and Head of Healthcare Investing, LongRiver Investments; and Dr. Maomeng Tong, Principal, INCE Capital, who shared strategic insights for biotechs pursuing cross‑border capital or collaboration.
"Asia's steady capital recovery is fueling more diverse, higher-quality biotech deals. Investors are increasingly favoring more scalable, capital-efficient, innovative business models. One such emerging approach is the NewCo model, which combines equity participation with experienced management teams, leverages Asia's efficient clinical speed and ecosystem partners to accelerate data generation, and enables program-level co-development. These elements de-risk global development in multiple ways, making the opportunities more attractive for international syndicates," said Dr. Alva Chen, Managing Director and Head of Therapeutics, VMS Group.
"Clinical development now spans multiple regions more than ever and requires an integrated strategy. HiRO's cross‑border capabilities across APAC, the US, and Europe allow us to align regulatory strategy, site selection, and operational execution to bridge data across regions, accelerate timelines, and reduce redundant costs. By leveraging local expertise, regional cost advantages, and selective partnerships, we help biotech sponsors generate earlier, higher‑quality readouts and deploy capital more efficiently to advance financing and licensing goals," said Dr. Karen Chu, Founder and CEO of HiRO.
About Harvest Integrated Research Organization (HiRO)
Harvest Integrated Research Organization (HiRO) is a globally oriented, innovative clinical research organization. With global operations and integrated capabilities, HiRO provides a full range of cross-border solutions and services to its clients, including early pre-clinical strategic planning, clinical trial design, regulatory affairs, pharmacovigilance, statistics, data management, end-to-end project management, and clinical and medical monitoring services.
As an emerging global CRO, HiRO strives to become a market-leading, integrated global clinical research organization that works collaboratively with biotech and pharmaceutical companies to bring new products from the laboratory to the market, providing more effective solutions for patients worldwide. For more information on HiRO, please visit www.harvestiro.com.
SOMERSET, N.J., March 3, 2026 /PRNewswire/ -- Harvest Integrated Research Organization (HiRO), a global contract research organization specializing in strategic planning for clinical development and cross‑border clinical trial solutions and services, concluded a successful presence at the 44th Annual J.P. Morgan Healthcare Conference in San Francisco.
HiRO's Founder and CEO, Dr. Karen Chu, was a featured speaker at RESI JPM, where she moderated a panel convening investors and strategics at the forefront of the cell and gene therapy revolution. Panelists Robert Balfour of ALSA Ventures and Bettina Ernst of BERNINA BioInvest highlighted the current momentum in cell and gene therapies, discussed how they assess technical and commercial risk, identified partnership models that accelerate progress, and outlined where capital is flowing in this rapidly advancing field.
Dr. Chu also led a workshop titled "Leveraging Asia: How to Navigate Asian VC Investment Mandates." The session opened with the 2026 industry outlook and key trends, then highlighted priority therapeutic areas. It explained the NewCo model and the criteria Asian venture capital firms use to evaluate global biotech assets, their co‑investment preferences, and expectations for commercialization. The workshop featured Asia‑based investors Dr. Alva Chen, Managing Director and Head of Therapeutics, VMS Group; Jayson Lee, Partner and Head of Healthcare Investing, LongRiver Investments; and Dr. Maomeng Tong, Principal, INCE Capital, who shared strategic insights for biotechs pursuing cross‑border capital or collaboration.
"Asia's steady capital recovery is fueling more diverse, higher-quality biotech deals. Investors are increasingly favoring more scalable, capital-efficient, innovative business models. One such emerging approach is the NewCo model, which combines equity participation with experienced management teams, leverages Asia's efficient clinical speed and ecosystem partners to accelerate data generation, and enables program-level co-development. These elements de-risk global development in multiple ways, making the opportunities more attractive for international syndicates," said Dr. Alva Chen, Managing Director and Head of Therapeutics, VMS Group.
"Clinical development now spans multiple regions more than ever and requires an integrated strategy. HiRO's cross‑border capabilities across APAC, the US, and Europe allow us to align regulatory strategy, site selection, and operational execution to bridge data across regions, accelerate timelines, and reduce redundant costs. By leveraging local expertise, regional cost advantages, and selective partnerships, we help biotech sponsors generate earlier, higher‑quality readouts and deploy capital more efficiently to advance financing and licensing goals," said Dr. Karen Chu, Founder and CEO of HiRO.
About Harvest Integrated Research Organization (HiRO)
Harvest Integrated Research Organization (HiRO) is a globally oriented, innovative clinical research organization. With global operations and integrated capabilities, HiRO provides a full range of cross-border solutions and services to its clients, including early pre-clinical strategic planning, clinical trial design, regulatory affairs, pharmacovigilance, statistics, data management, end-to-end project management, and clinical and medical monitoring services.
As an emerging global CRO, HiRO strives to become a market-leading, integrated global clinical research organization that works collaboratively with biotech and pharmaceutical companies to bring new products from the laboratory to the market, providing more effective solutions for patients worldwide. For more information on HiRO, please visit www.harvestiro.com.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

HiRO Leads Cross‑Border Dialogue at JPM, Offering Insights for Biotechs Seeking Asian Investment