The Southern Europe's leading food business platform will connect Italian and international producers with the Japanese growing market
MILAN, March 31, 2026 /PRNewswire/ -- 90 Japanese buyers, representing leading companies in the country's agri-food sector, have been confirmed to attend TUTTOFOOD, Southern Europe's leading food business platform, taking place in Milan from 11 to 14 May 2026. The trade fair will provide a high-level platform to foster business development, offering them an unmissable opportunity to expand their agri-food market by strengthening commercial partnerships with Italian exhibitors.
Imports are a dominant component of Japan's agri-food trade. Their value rises from 6,378 billion yen in 2016 to 10,133 billion yen in 2025, marking an overall increase of approximately 59%. This underscores the critical role of international suppliers, including Italy, in meeting Japan's domestic demand for high-quality food and beverage products.
According to a market note from ITA – Italian Trade Agency, Tokyo Office, the Food & Beverage sector in Japan is valued at approximately USD 560 billion (combined retail and food service estimates). Demand for "Made in Italy" products has never been higher in terms of value. With around 60% of consumed food being imported, Japan represents one of the largest markets for foreign food products.
Consumption patterns in Japan are increasingly polarized: while the mid-range segment is contracting, the premium segment continues to expand at an average rate of 5% annually, driven by consumers seeking excellence, authenticity, and strong product narratives. At the same time, the recovery of the HoReCa channel—supported by a rebound in tourism, with over 30 million visitors—has significantly boosted demand for high-quality Italian ingredients, particularly within the fine dining and upscale restaurant segment.
The increase in imports is not driven solely by quantitative needs, but reflects a demand increasingly focused on quality, diversity, and ingredients for international cuisines. In this context, the Japanese market remains structurally open and receptive to foreign agri-food supplies, particularly in higher-value segments, while maintaining a strong focus on premium positioning.
Antonio Cellie, CEO of Fiere di Parma, said: "There is a growing demand in Japan for high-quality Italian products, highlighting Japan's evolution into a strategic, long-term partner while sharing Italy's commitment to quality, traceability, and culinary excellence. TUTTOFOOD represents a critical lever in this context, enabling high-quality business interactions with Italian distributors, retailers, and importers, while also giving insights on consumer behaviour and market trends".
TUTTOFOOD plays a key role as enablers of global business development, by offering a structured environments for direct engagement between producers and qualified buyers and act as a catalyst for innovation and international expansion.
The upcoming edition is expected to feature a strong international footprint, with more than 30% of exhibitors from overseas and an exceptionally rich Buyers Program organized in cooperation with ITA - Italian Trade Agency, bringing together more than 4,000 top national and international professionals. Particular attention will be dedicated to the Asia-Pacific region, in line with its increasing relevance.
In this perspective, Japan and Italy play as strategic partners within a broader framework of mutual value creation. Shared principles—such as product quality, cultural heritage, and reliability—continue to underpin the relationship between Italian producers and Japanese buyers, creating a solid foundation for long-term collaboration and future opportunities.
The strategic ties between the two countries took center stage during a presentation event held at the Italian Embassy in Tokyo, as part of TUTTOFOOD 2026 international roadshow.
Photo: https://mma.prnasia.com/media2/2945630/Antonio_Cellie_CEO_Fiere_di_Parma.jpg?p=medium600
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

TUTTOFOOD 2026 CONNECTS GLOBAL AGRI-FOOD SUPPLIERS WITH JAPAN
|
In a separate announcement made today, the Company also secured A$83 million in private placement funding to support growth initiatives, including European expansion.
MELBOURNE, Australia, March 31, 2026 /PRNewswire/ -- 4DMedical Limited (ASX:4DX), the global leader in cardiothoracic imaging software, announces that its latest imaging technology, CT:VQ™ has received CE Mark certification for commercial use in the European Union. 4DMedical will quickly launch commercial deployment of CT:VQ across one of the world's largest respiratory imaging markets.
In a separate but concurrent announcement, 4DMedical has secured firm commitments from select institutional investors for an AU$83 million single-tranche private placement. This additional capital will support the Company's growth strategy, particularly the expansion of CT:VQ™ in Europe.
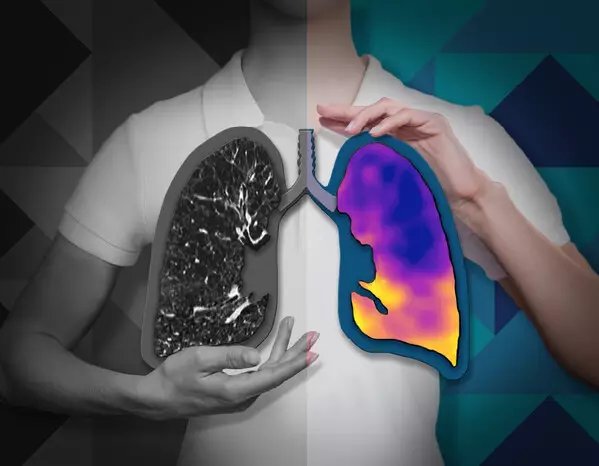
CT:VQ™ is the world's first and only non-contrast, ventilation-perfusion imaging technology, providing quantitative functional lung insights from routine non-contrast CT scans without the need for radiotracers or specialised nuclear medicine infrastructure.
Pulmonary disease is fundamentally functional—defined by abnormalities in ventilation and perfusion—yet most routine imaging available to radiologists and pulmonologists remains purely structural. CT:VQ™ bridges that gap by generating ventilation and perfusion maps allowing clinicians to visualize regional lung function with the anatomical clarity of CT.
For large medical institutions, CT:VQ™ is designed to fit within existing CT-based workflows and infrastructure. CT:VQ offers relief from long-standing constraints associated with conventional nuclear VQ imaging, including radiotracer availability, workforce limitations and operational complexity.
The European market represents a significant opportunity for advanced cardiothoracic imaging. With a population of more than 450 million, a highly developed hospital-based imaging infrastructure and an extensive installed base of CT scanners, the EU is well positioned for adoption of CT-based functional lung imaging. 4DMedical estimates that approximately 400,000 nuclear VQ scans are performed annually across the EU, underscoring the scale of clinical need.
"CE Mark certification for CT:VQ™ is a significant milestone that opens access to one of the world's largest and most sophisticated healthcare markets," said Andreas Fouras, Managing Director, CEO and Founder of 4DMedical. "Combined with FDA clearance, 4DMedical now has regulatory clearance to rapidly commercialise CT:VQ™ across both the U.S. and the EU.
"The clinical need for CT:VQ™ is universal. The limitations of nuclear VQ scanning—radiotracer constraints, limited access and operational complexity—exist across healthcare systems globally. Europe has the clinical expertise, imaging infrastructure and research leadership to play a major role in the adoption and evidence generation for this technology."
The CE Mark follows growing U.S. momentum for CT:VQ™, which is now deployed at six leading academic medical centres: Stanford, Cleveland Clinic, University of Miami, UC San Diego Health, University of Chicago Medicine, and the recently announced initial deployment at Mayo Clinic. 4DMedical believes this early adoption by major U.S. institutions demonstrates strong interest in CT:VQ™ among leading radiology and pulmonary teams and provides a blueprint for expansion into Europe.
4DMedical said it is now advancing plans to expand commercial engagement across Europe and to work with leading hospitals and clinicians on adoption, evaluation and research initiatives for CT:VQ™.
About CT:VQ™
CT:VQ™ is 4DMedical's proprietary ventilation-perfusion imaging solution that generates functional lung information from non-contrast CT scans. Built on the Company's patented XV Technology®, CT:VQ™ is designed to provide quantitative ventilation and perfusion insights within familiar CT workflows, without requiring radiotracers.
About 4DMedical
4DMedical Limited (ASX:4DX) is a global medical technology company that creates and deploys the most advanced software-based cardiothoracic imaging technology—integrating both proprietary algorithms and artificial intelligence. 4DMedical's software platform delivers deep, quantitative, insights from routine clinical imaging to help clinicians assess lung function and cardiopulmonary disease.
These insights help respiratory physicians, radiologists and hospital teams quantify regional lung function, support diagnosis and disease monitoring, and inform treatment and surgical planning. 4DMedical's solutions are designed to integrate into existing clinical workflows. They provide actionable, patient-specific information that supports better decision-making and operational efficiency across a range of respiratory conditions. Learn more at www.4dmedical.com.
Photo - https://mma.prnasia.com/media2/2944827/Lungs_Graphic_Cutouts_Color_BW.jpg?p=medium600
Logo - https://mma.prnasia.com/media2/2730645/5888947/4DMedical_Logo.jpg?p=medium600
In a separate announcement made today, the Company also secured A$83 million in private placement funding to support growth initiatives, including European expansion.
MELBOURNE, Australia, March 31, 2026 /PRNewswire/ -- 4DMedical Limited (ASX:4DX), the global leader in cardiothoracic imaging software, announces that its latest imaging technology, CT:VQ™ has received CE Mark certification for commercial use in the European Union. 4DMedical will quickly launch commercial deployment of CT:VQ across one of the world's largest respiratory imaging markets.
In a separate but concurrent announcement, 4DMedical has secured firm commitments from select institutional investors for an AU$83 million single-tranche private placement. This additional capital will support the Company's growth strategy, particularly the expansion of CT:VQ™ in Europe.
CT:VQ™ is the world's first and only non-contrast, ventilation-perfusion imaging technology, providing quantitative functional lung insights from routine non-contrast CT scans without the need for radiotracers or specialised nuclear medicine infrastructure.
Pulmonary disease is fundamentally functional—defined by abnormalities in ventilation and perfusion—yet most routine imaging available to radiologists and pulmonologists remains purely structural. CT:VQ™ bridges that gap by generating ventilation and perfusion maps allowing clinicians to visualize regional lung function with the anatomical clarity of CT.
For large medical institutions, CT:VQ™ is designed to fit within existing CT-based workflows and infrastructure. CT:VQ offers relief from long-standing constraints associated with conventional nuclear VQ imaging, including radiotracer availability, workforce limitations and operational complexity.
The European market represents a significant opportunity for advanced cardiothoracic imaging. With a population of more than 450 million, a highly developed hospital-based imaging infrastructure and an extensive installed base of CT scanners, the EU is well positioned for adoption of CT-based functional lung imaging. 4DMedical estimates that approximately 400,000 nuclear VQ scans are performed annually across the EU, underscoring the scale of clinical need.
"CE Mark certification for CT:VQ™ is a significant milestone that opens access to one of the world's largest and most sophisticated healthcare markets," said Andreas Fouras, Managing Director, CEO and Founder of 4DMedical. "Combined with FDA clearance, 4DMedical now has regulatory clearance to rapidly commercialise CT:VQ™ across both the U.S. and the EU.
"The clinical need for CT:VQ™ is universal. The limitations of nuclear VQ scanning—radiotracer constraints, limited access and operational complexity—exist across healthcare systems globally. Europe has the clinical expertise, imaging infrastructure and research leadership to play a major role in the adoption and evidence generation for this technology."
The CE Mark follows growing U.S. momentum for CT:VQ™, which is now deployed at six leading academic medical centres: Stanford, Cleveland Clinic, University of Miami, UC San Diego Health, University of Chicago Medicine, and the recently announced initial deployment at Mayo Clinic. 4DMedical believes this early adoption by major U.S. institutions demonstrates strong interest in CT:VQ™ among leading radiology and pulmonary teams and provides a blueprint for expansion into Europe.
4DMedical said it is now advancing plans to expand commercial engagement across Europe and to work with leading hospitals and clinicians on adoption, evaluation and research initiatives for CT:VQ™.
About CT:VQ™
CT:VQ™ is 4DMedical's proprietary ventilation-perfusion imaging solution that generates functional lung information from non-contrast CT scans. Built on the Company's patented XV Technology®, CT:VQ™ is designed to provide quantitative ventilation and perfusion insights within familiar CT workflows, without requiring radiotracers.
About 4DMedical
4DMedical Limited (ASX:4DX) is a global medical technology company that creates and deploys the most advanced software-based cardiothoracic imaging technology—integrating both proprietary algorithms and artificial intelligence. 4DMedical's software platform delivers deep, quantitative, insights from routine clinical imaging to help clinicians assess lung function and cardiopulmonary disease.
These insights help respiratory physicians, radiologists and hospital teams quantify regional lung function, support diagnosis and disease monitoring, and inform treatment and surgical planning. 4DMedical's solutions are designed to integrate into existing clinical workflows. They provide actionable, patient-specific information that supports better decision-making and operational efficiency across a range of respiratory conditions. Learn more at www.4dmedical.com.
Photo - https://mma.prnasia.com/media2/2944827/Lungs_Graphic_Cutouts_Color_BW.jpg?p=medium600
Logo - https://mma.prnasia.com/media2/2730645/5888947/4DMedical_Logo.jpg?p=medium600
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
4DMedical receives CE Mark for CT:VQ™, enabling commercial launch across the European Union











