Highlights
- Median Tmax of 10 minutes for SPONTAN® (5 mg) versus 60 minutes for oral vardenafil (20mg), consistent with Phase I study.
- Interim data provide the key pharmacokinetic dataset requested by the FDA, including rapid absorption, repeat-dose profile, with no drug accumulation observed, and consistent pharmacokinetics across adult and geriatric populations.
- Pharmacokinetic profile in subjects ≥65 years comparable to adults in the dataset, addressing a key FDA Pre-IND requirement.
- Consistent safety profile observed, with no serious adverse events, no Grade 3 or 4 treatment-emergent adverse events, and no treatment-related discontinuations.
- No drug accumulation observed following five days of repeat intranasal dosing (5 mg).
- Interim data are expected to inform the Company's planned FDA 505(b)(2) regulatory pathway.
- Final results, including full statistical analysis, expected Q3 CY2026.
SYDNEY, May 4, 2026 /PRNewswire/ -- LTR Pharma Limited (ASX:LTP) ("LTR Pharma" or "the Company") reports interim Phase II pharmacokinetic (PK) and safety data for SPONTAN®, demonstrating a rapid time to peak concentration (median Tmax 10 minutes) and a consistent safety profile, with no serious or severe treatment-emergent adverse events observed. The data support the Company's planned FDA 505(b)(2) regulatory pathway, subject to completion of final statistical analysis.
These results indicate a pharmacokinetic profile distinct from oral PDE5 inhibitors, consistent with rapid-onset, on-demand use.
The interim results indicate that the Study has characterised the key pharmacokinetic parameters requested by the FDA, including rapid absorption, repeat-dose behaviour, and consistency across adult and geriatric populations, subject to final statistical analysis.
The treatment phase of the Study has been completed, and a preliminary analysis of the data has been undertaken. The analysis demonstrates a rapid-onset pharmacokinetic profile and favourable safety in both adult and geriatric populations. The Study was designed in accordance with FDA Pre-IND guidance and supports LTR Pharma's planned 505(b)(2) development pathway.
Statistical analysis of the dataset is ongoing. Full results, including final pharmacokinetic, safety, and dose-proportionality analyses, are expected to be released in Q3 CY2026.
Key Metrics: Interim Phase II Pharmacokinetic Data
| Metric | SPONTAN® 2.5 mg Nasal Spray | SPONTAN® 5 mg Nasal Spray | vardenafil 20 mg Oral Tablet |
| Trial population (n) | 27 (incl. 14 ≥65 years) | 27 (incl. 14 ≥65 years) | 27 (incl. 14 ≥65 years) |
| Cmax (ng/mL), Trial population | 5.8 ± 4.9 | 8.9 ± 6.8 | 19.8 ± 8.7 |
| Cmax (ng/mL), Geriatric (≥65) | 5.3 ± 5.0 | 7.3 ± 5.5 | 16.6 ± 7.8 |
| Tmax (minutes), median (range), Trial population | 10 (5–60) | 10 (10–15) | 60 (30–180) |
| Tmax (minutes), median (range), Geriatric (≥65) | 15 (10–60) | 10 (10–15) | 60 (30–180) |
| Accumulation Ratio (AUC₀–24 D10/AUC₀–24 D14) | n.a. | 1.0 ± 0.9 | n.a. |
| Serious Adverse Events | 0 | 0 | 0 |
| Grade 3 or 4 Treatment-Emergent Adverse Events | 0 | 0 | 0 |
| Treatment-related discontinuations | 0 | 0 | 0 |
Metric
SPONTAN® 2.5 mg Nasal Spray
SPONTAN® 5 mg Nasal Spray
vardenafil 20 mg Oral Tablet
Trial population (n)
27 (incl. 14 ≥65 years)
27 (incl. 14 ≥65 years)
27 (incl. 14 ≥65 years)
Cmax (ng/mL), Trial population
5.8 ± 4.9
8.9 ± 6.8
19.8 ± 8.7
Cmax (ng/mL), Geriatric (≥65)
5.3 ± 5.0
7.3 ± 5.5
16.6 ± 7.8
Tmax (minutes), median (range), Trial population
10 (5–60)
10 (10–15)
60 (30–180)
Tmax (minutes), median (range), Geriatric (≥65)
15 (10–60)
10 (10–15)
60 (30–180)
Accumulation Ratio (AUC₀–24 D10/AUC₀–24 D14)
n.a.
1.0 ± 0.9
n.a.
Serious Adverse Events
Grade 3 or 4 Treatment-Emergent Adverse Events
Treatment-related discontinuations
The data are interim and based on 27 subjects. Final statistical analysis remains ongoing. Cmax values expressed as mean ± standard deviation. n.a. = not applicable. Observed lower Cmax relative to oral vardenafil is consistent with the lower dose, while maintaining rapid systemic exposure.
Study Findings
The treatment phase of the SPONTAN Phase II clinical study has been completed, and a preliminary analysis of the data has been undertaken. The primary objectives of the Study, as agreed with the FDA, were to characterise single- and multiple-dose pharmacokinetics of intranasally delivered vardenafil (SPONTAN), to evaluate the effect of intranasal delivery on drug accumulation following repeat dosing, and to assess pharmacokinetic differences between adult and geriatric populations.
Onset of action. SPONTAN (5 mg vardenafil) demonstrated a median Tmax of 10 minutes, compared with a median Tmax of 60 minutes for the 20 mg vardenafil oral tablet. The Tmax range for SPONTAN (5 mg) was 10–15 minutes, compared with 30–180 minutes for the oral tablet.
Geriatric profile. Pharmacokinetic profiles in subjects ≥65 years were comparable to those in the adult cohort, with no apparent differences observed in the dataset, and the median Tmax of 10 minutes for SPONTAN (5 mg) was maintained across both age groups. These findings address a key FDA requirement and may support use across age groups.
Drug accumulation. Intranasal administration of SPONTAN (5 mg) over five consecutive days did not result in drug accumulation, with an accumulation ratio of 1.0 ± 0.9, consistent with the multiple-dose study objectives agreed with the FDA.
Safety and tolerability. Preliminary safety analysis did not identify any new or unexpected treatment-emergent adverse events. Across all three dose groups, no serious adverse events, no Grade 3 or 4 treatment-emergent adverse events, and no treatment-related discontinuations were observed in the interim dataset.
Clinical and Commercial Relevance
Oral PDE5 inhibitors require advance planning, with onset times ranging from approximately 30 minutes to over 2 hours. Dropout rates exceeding 50% have been reported in the published literature, with lack of spontaneity a consistently cited reason for discontinuation.[1]
The data indicate a rapid-onset profile relative to oral PDE5 inhibitors, which may be relevant for patient adherence given reported discontinuation rates associated with delayed onset. Consistent pharmacokinetics in the geriatric cohort may be relevant across age groups, subject to final analysis and regulatory review.
Regulatory Pathway
The Phase II PK Study was specifically designed in accordance with FDA Pre-IND guidance and is a key requirement in LTR Pharma's FDA 505(b)(2) development pathway. The data are expected to inform LTR Pharma's planned regulatory strategy, including the proposed 505(b)(2) submission in the United States, subject to completion of final analysis and regulatory feedback.
Data Status and Limitations
The data reported in this announcement are based on 27 subjects who completed the dosing phase of the Phase II PK Study and reflect a preliminary analysis. Final statistical analysis, including dose proportionality, full pharmacokinetic modelling, and the complete safety dataset, remains ongoing. The final dataset and statistical conclusions may differ from the interim observations reported. Full study results are expected to be released in Q3 CY2026.
Study Design
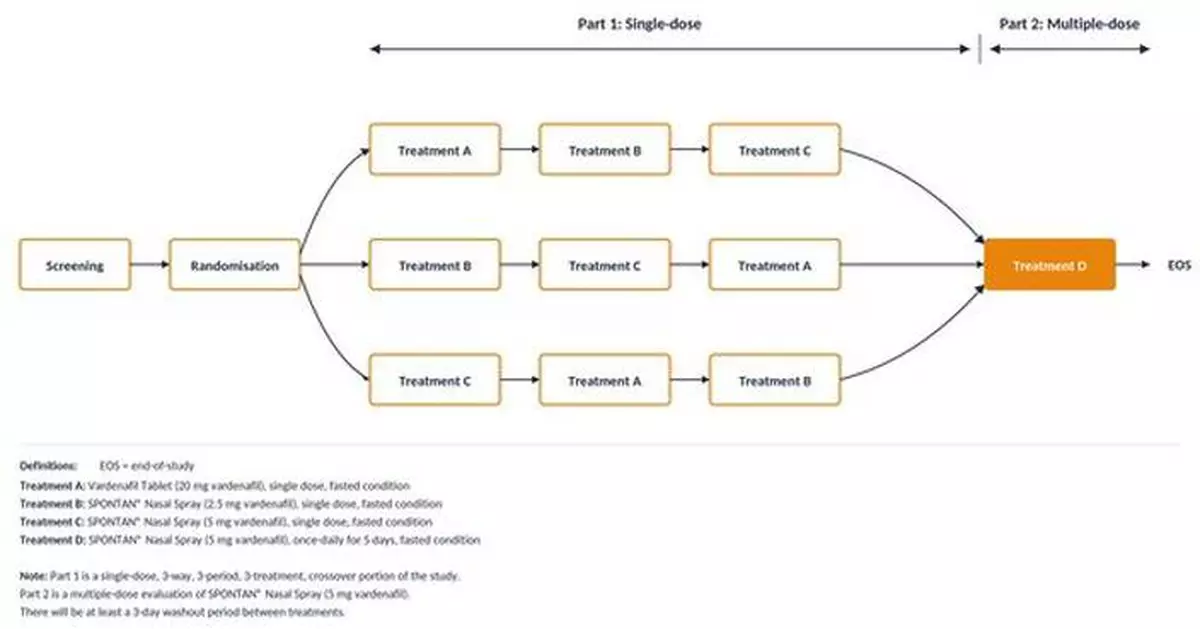
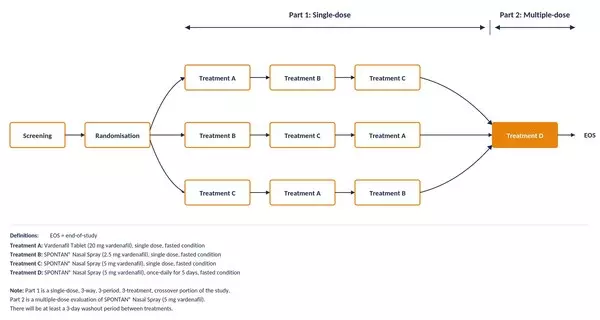
Figure 1: SPONTAN Phase II study design. Randomised, three-way crossover (Part 1) followed by multiple-dose evaluation (Part 2).
| Parameter | Detail |
| Study reference | SDS089 (SPONTAN)-PK-02 |
| Phase | Phase II |
| Design | Randomised, open-label, single- and multiple-dose, three-way crossover |
| Number of subjects | 27 healthy adult male subjects enrolled, including 14 subjects aged ≥65 years |
| Comparator | vardenafil 20 mg oral tablet |
| SPONTAN doses | 2.5 mg and 5 mg vardenafil intranasal |
| Part 1 | Single-dose, three-period, three-way crossover (treatment sequences ABC, BCA, CAB), separated by a 3-day washout |
| Part 2 | Multiple-dose study: 5 daily doses of SPONTAN 5 mg over 5 days |
| Setting | Confinement to the clinical research unit for a minimum of 15 nights |
| Primary endpoints | Single- and multiple-dose pharmacokinetics of vardenafil and metabolite M1; dose proportionality |
| Secondary endpoints | Effect of age (<65 vs ≥65 years) on PK; intranasal local safety and tolerability |
| Clinical site | Scientia Clinical Research, Sydney |
| Clinical Research Organisation | Southern Star Research |
| Bioanalytical | Resolian Bioanalytics |
Parameter
Detail
Study reference
SDS089 (SPONTAN)-PK-02
Phase
Phase II
Design
Randomised, open-label, single- and multiple-dose, three-way crossover
Number of subjects
27 healthy adult male subjects enrolled, including 14 subjects aged ≥65 years
Comparator
vardenafil 20 mg oral tablet
SPONTAN doses
2.5 mg and 5 mg vardenafil intranasal
Part 1
Single-dose, three-period, three-way crossover (treatment sequences ABC, BCA, CAB), separated by a 3-day washout
Part 2
Multiple-dose study: 5 daily doses of SPONTAN 5 mg over 5 days
Setting
Confinement to the clinical research unit for a minimum of 15 nights
Primary endpoints
Single- and multiple-dose pharmacokinetics of vardenafil and metabolite M1; dose proportionality
Secondary endpoints
Effect of age (<65 vs ≥65 years) on PK; intranasal local safety and tolerability
Clinical site
Scientia Clinical Research, Sydney
Clinical Research Organisation
Southern Star Research
Bioanalytical
Resolian Bioanalytics
The Study design, including endpoint selection and the inclusion of a robust geriatric cohort, was agreed upon with the FDA during the Pre-IND meeting in 2025.
LTR Pharma Executive Chairman, Lee Rodne, said:
"The interim Phase II data are consistent with our Phase I findings and reinforce SPONTAN's differentiated rapid-onset profile, including in men aged 65 and over, a group that often requires dose adjustment with oral PDE5 therapies. Importantly, no serious adverse events, no severe treatment-emergent events, and no treatment-related discontinuations were observed. This Study was specifically designed in accordance with FDA Pre-IND guidance, and we are now focused on completing the statistical analysis and progressing our 505(b)(2) regulatory strategy in the United States."
Next Steps
LTR Pharma, in collaboration with its Clinical Research Organisation, will complete the final statistical analysis over the coming months, with full Study results expected to be released in Q3 CY2026. The complete dataset will be used to support LTR Pharma's planned FDA 505(b)(2) submission and ongoing regulatory engagement in Australia, the United States, and other key markets.
This announcement has been approved by the Board of Directors.
About LTR Pharma
LTR Pharma is a commercial-stage pharmaceutical company delivering innovative therapies to address significant unmet medical needs through its proprietary intranasal drug-delivery platform. The Company has successfully commercialised its rapid-acting treatment technology in Australia and is expanding access whilst advancing regulatory pathways in the US and other key markets.
LTR's lead products, SPONTAN® and ROXUS®, are fast-acting intranasal sprays for the treatment of erectile dysfunction, enabling onset of action in 10 minutes or less. Building on this proven technology, the Company is now advancing OROFLOW®, a novel intranasal spray under development for the treatment of Oesophageal Motility Disorders (OMD) – a debilitating group of conditions affecting swallowing function.
Through strategic partnerships, LTR Pharma is expanding its pipeline and global footprint to deliver differentiated, patient-centric treatments that enhance quality of life.
LTR Pharma Investor Centre
Stay informed with LTR Pharma's latest announcements and market updates by visiting our Investor Centre.
| [1] Carvalheira et al., "Dropout in the Treatment of Erectile Dysfunction with PDE5: A Study on Predictors and a Qualitative Analysis of Reasons for Discontinuation," Journal of Sexual Medicine, May 2012. |
[1] Carvalheira et al., "Dropout in the Treatment of Erectile Dysfunction with PDE5: A Study on Predictors and a Qualitative Analysis of Reasons for Discontinuation," Journal of Sexual Medicine, May 2012.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
SPONTAN® Phase II interim data demonstrates rapid onset and pharmacokinetic profile addressing FDA Pre-IND requirements across adult and ≥65 populations