SAN FRANCISCO--(BUSINESS WIRE)--Feb 26, 2025--
Loyal, a clinical-stage animal health company developing longevity drugs for dogs, announced today that the FDA’s Center for Veterinary Medicine has accepted the Reasonable Expectation of Effectiveness (RXE) section of its conditional approval application for LOY-002. The drug aims to extend the lifespan of senior dogs and maintain their quality of life as they age, building on the company’s previous RXE acceptance for a longevity drug specifically targeting the short lifespan of large breed dogs.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250226676005/en/
The company also announced it has raised a $22M million B-2 round from Valor Equity Partners and Collaborative Fund, among others, in addition to its $45M Series B in 2024. This brings total investment in Loyal’s to over $150M.
“Everything we do is in service of helping dogs live longer, healthier lives,” said Loyal Founder and CEO Celine Halioua. “These two milestones represent our ongoing commitment to that mission through years of diligence and hard work. Proving efficacy is one of the most challenging parts of developing a novel drug. While we still have significant work to do, RXE increases the probability that dogs will soon have access to our longevity drugs.”
A New Preventive Paradigm
The FDA's Center for Veterinary Medicine has determined that Loyal's data provides reasonable expectation of LOY-002's intended effect of extending canine lifespan, marking a significant milestone in the company's pursuit of conditional approval.
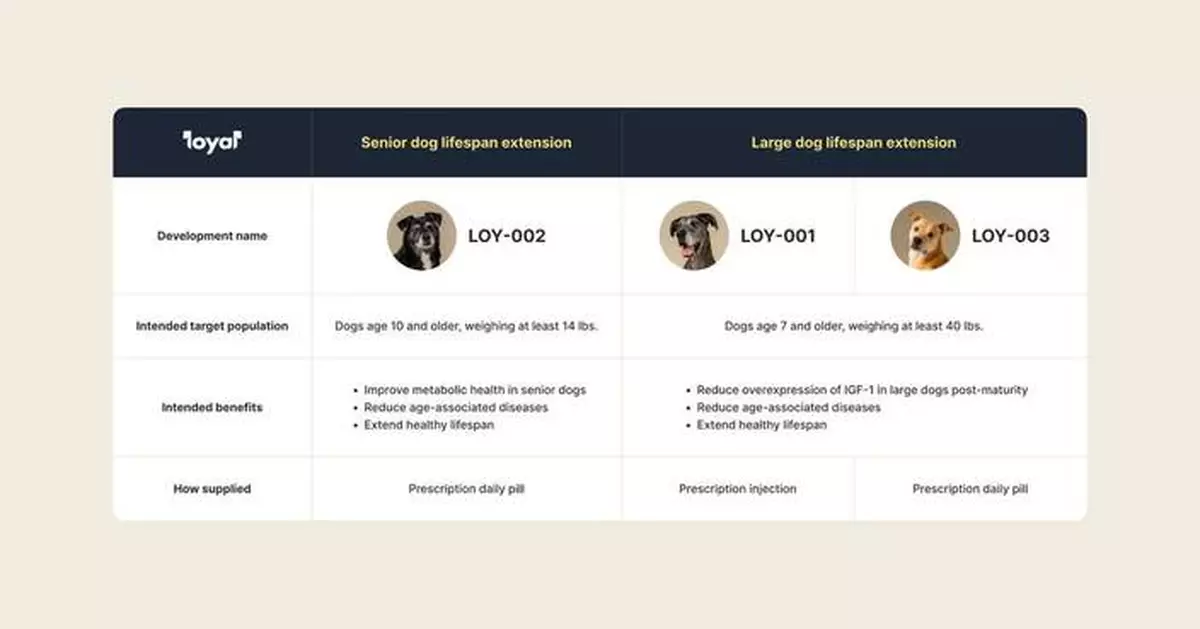
LOY-002, a daily flavored tablet developed for dogs aged 10 and older weighing at least 14 pounds, represents a groundbreaking approach to canine longevity by targeting metabolic dysfunction, one of the key underlying mechanisms of aging. This preventive therapy aims to reduce the incidence and severity of age-associated diseases that diminish senior dogs' quality of life, offering a practical alternative to established longevity interventions like caloric restriction.
The FDA's acceptance of RXE demonstrates confidence in Loyal's innovative approach, and the company anticipates completion of the manufacturing and safety requirements for Expanded Conditional Approval (XCA) from the FDA by the end of 2025.
“Loyal’s progress in bringing their products to market continues to impress, especially since they are breaking new regulatory ground,” said Dr. Linda Rhodes, VMD, PhD, a veterinarian and experienced animal health executive who serves on Loyal’s Board of Directors.
As the company pursues its XCA application, Loyal is simultaneously running its groundbreaking clinical trial, the STAY study. The STAY study is LOY-002’s pivotal efficacy study that dosed its first dog in December 2023 and will take place over approximately four years. It is the first and only FDA-concurred clinical trial for longevity and is the largest veterinary trial in history, recruiting over 1,000 dogs from 70 veterinary clinics across the country.
“Achieving RXE acceptance is a testament to the ongoing importance of the STAY study,” said Dr. Ellen Ratcliff, VP of Clinical and Veterinary Medicine at Loyal. “Completing enrollment and seeing what the data shows over the next several years is critical, but this is an important step in the process. We now have validation from one of the highest quality regulatory agencies in the world that the drug we’re testing has a reasonable expectation of extending a dog’s life.”
B-2 Fundraising Round
In addition to achieving RXE for its second drug program, Loyal successfully completed a $22M B-2 funding round, led by Valor Equity Partners and Collaborative Fund, among others. Along with his investment in Loyal, Jonathan Shulkin, Partner and Co-President at Valor Equity Partners, is joining the company’s Board of Directors.
This raise represents a significant premium on the Loyal’s Series B last year and will support the company’s execution of key go-to-market efforts to bring LOY-002 to veterinarians and dog owners soon, in addition to advancing the other drugs in development across its portfolio: LOY-001 and LOY-003, which are designed to extend the healthy lifespan of large-breed dogs.
About Loyal
Loyal is an animal health company developing the first drugs intended to help dogs live longer, healthier lives. By targeting the underlying mechanisms of aging, Loyal hopes to extend the lifespan of dogs and improve their quality of life as they age. Loyal’s work is based on decades of research, and their team of experts in dog health and longevity is dedicated to furthering this research and developing better ways to quantify and improve the aging process in dogs. It currently has three drugs in development and is making progress toward approval from the FDA’s Center for Veterinary Medicine.
For more information, please visit loyal.com.
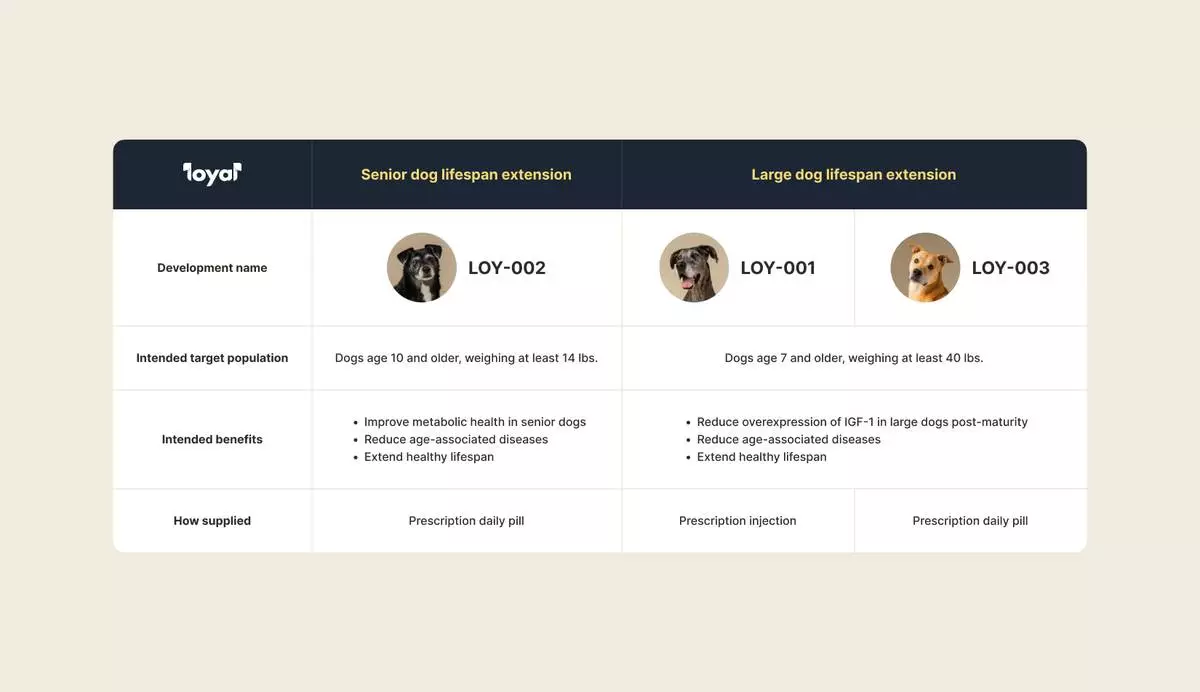

An overview of Loyal's three drugs in development: LOY-001, LOY-002, and LOY-003 (Photo: Business Wire)