TARRYTOWN, N.Y.--(BUSINESS WIRE)--Jul 24, 2025--
Siemens Healthineers has earned My Green Lab ACT Ecolabel Certification for more than 150 immunoassay and clinical chemistry reagents used globally by medical laboratories for in vitro diagnostic testing. 1 The company is the first to earn certification for both reagents and analyzers in its Atellica portfolio. Notable assays with the certification include its High-Sensitivity Troponin I assay, which is the first available assay in the U.S. to aid heart attack diagnosis and aid in identifying future risk of death and cardiac events, and its Enhanced Liver Fibrosis (ELF) Test, the first prognostic test used to help assess the likelihood of disease progression in patients with advanced fibrosis due to metabolic dysfunction-associated steatohepatitis (MASH). 2
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20250724568645/en/
The reagents were rigorously evaluated and independently certified through My Green Lab’s comprehensive assessment process. This globally recognized standard provides transparent, third-party verified environmental impact data for laboratory consumables, chemicals, and equipment. The ACT Ecolabel evaluates products across key environmental impact categories—including manufacturing practices, energy and water consumption, chemicals, waste, and packaging—and distills this information into a total score that helps scientists and procurement experts better evaluate the sustainability of products for their healthcare facilities.
"Resilient performance and environmental responsibility matter to labs of all sizes," said Lisa Rose, head of core laboratory solutions for Diagnostics at Siemens Healthineers. "Our assays in combination with our instruments offer unparalleled testing solutions to empower laboratories to run their labs more sustainably." The Atellica analyzers support sustainable lab operations, having demonstrated up to 48% energy reduction in reported use cases. 3
"As the leading laboratory sustainability certification organization in life sciences and pharma, My Green Lab is thrilled to combine our certification expertise with Siemens Healthineers’ innovative medical technology solutions to create a powerful pathway for clinical laboratories worldwide to reduce their environmental footprint while maintaining exceptional diagnostic quality," said James Connelly, CEO of My Green Lab.
In its continual pursuit of more sustainable IVD manufacturing, Siemens Healthineers also now ships specialty laboratory reagents with new packaging that reduces volume and replaces plastic inlays with cardboard, expected to reduce 39 annual tons of polystyrene plastics and saving 161 tons of CO 2— the equivalent weight of 20 cars and annual energy use of 18 average U.S. homes.
For its achievements in sustainability, transparency, and product stewardship, Siemens Healthineers was recognized with a Sustainability Award at the 2025 Breakthroughs Conference and Exhibition—one of the leading and most influential U.S. healthcare conferences—by Premier, Inc., a healthcare improvement company that provides solutions to two-thirds of all providers in the U.S. The ELF Test also was recognized as a breakthrough immunoassay innovation.
More information about Siemens Healthineers clinical laboratory sustainability efforts can be found here.
1 The ‘My Green Lab’ and ‘ACT’ registered trademarks are owned and controlled by My Green Lab Inc.
2 Product claims and availability may vary by country.
3 Siemens Healthineers Shape 24 Spotlight Together towards a sustainable tomorrow. The outcomes achieved by Siemens Healthineers customers were achieved in the customers’ unique settings. Since there is no typical hospital or laboratory, and many variables exist (e.g., hospital/laboratory size, case mix, level of IT adoption), there can be no guarantee that others will achieve the same results.
Siemens Healthineers pioneers breakthroughs in healthcare. For everyone. Everywhere. Sustainably. The company is a global provider of healthcare equipment, solutions and services, with activities in more than 180 countries and direct representation in more than 70. The group comprises Siemens Healthineers AG, listed as SHL in Frankfurt, Germany, and its subsidiaries. As a leading medical technology company, Siemens Healthineers is committed to improving access to healthcare for underserved communities worldwide and is striving to overcome the most threatening diseases. The company is principally active in the areas of imaging, diagnostics, cancer care and minimally invasive therapies, augmented by digital technology and artificial intelligence. In fiscal 2024, which ended on September 30, 2024, Siemens Healthineers had approximately 72,000 employees worldwide and generated revenue of around €22.4 billion. Further information is available at www.siemens-healthineers.com.
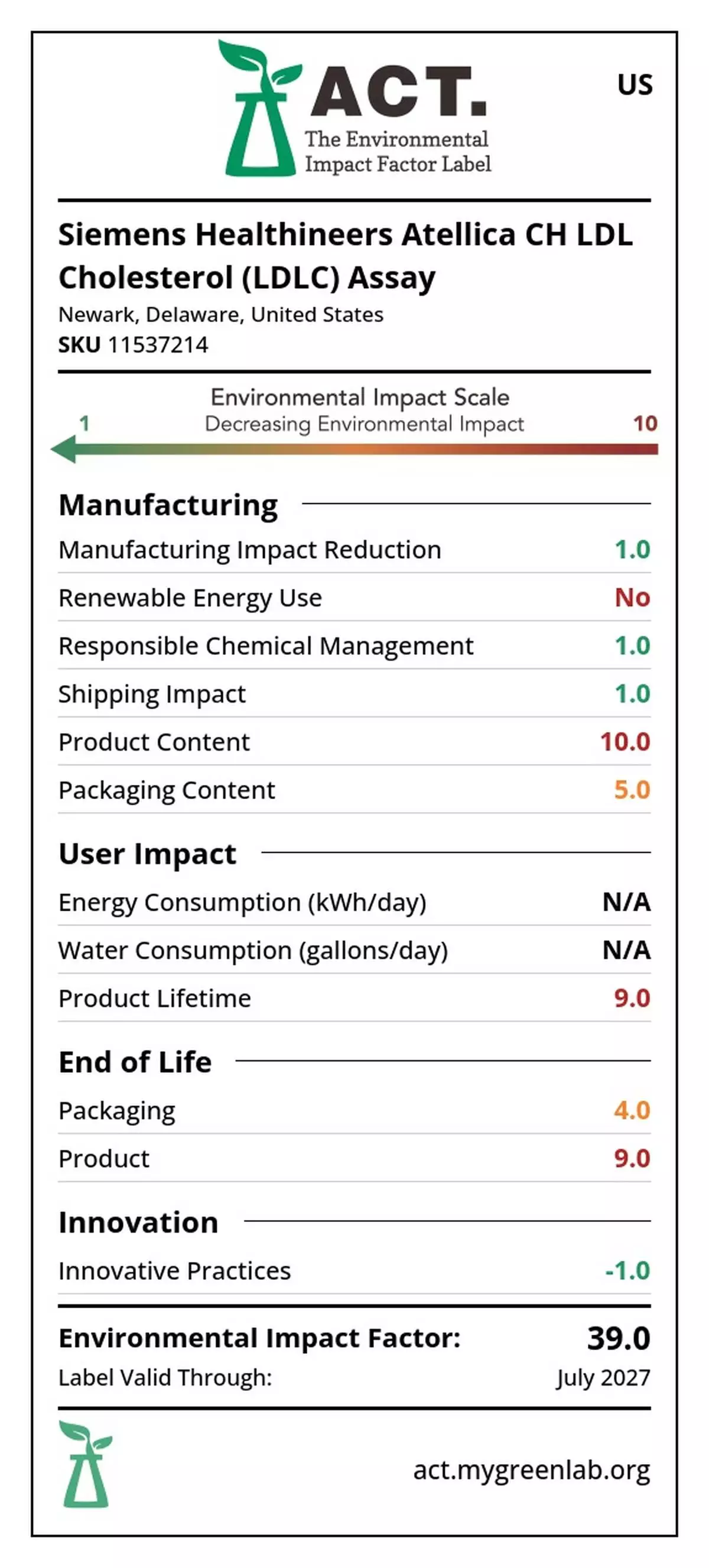

Siemens Healthineers has earned My Green Lab ACT Ecolabel Certification for more than 150 immunoassay and clinical chemistry reagents used globally by medical laboratories for in vitro diagnostic testing.










