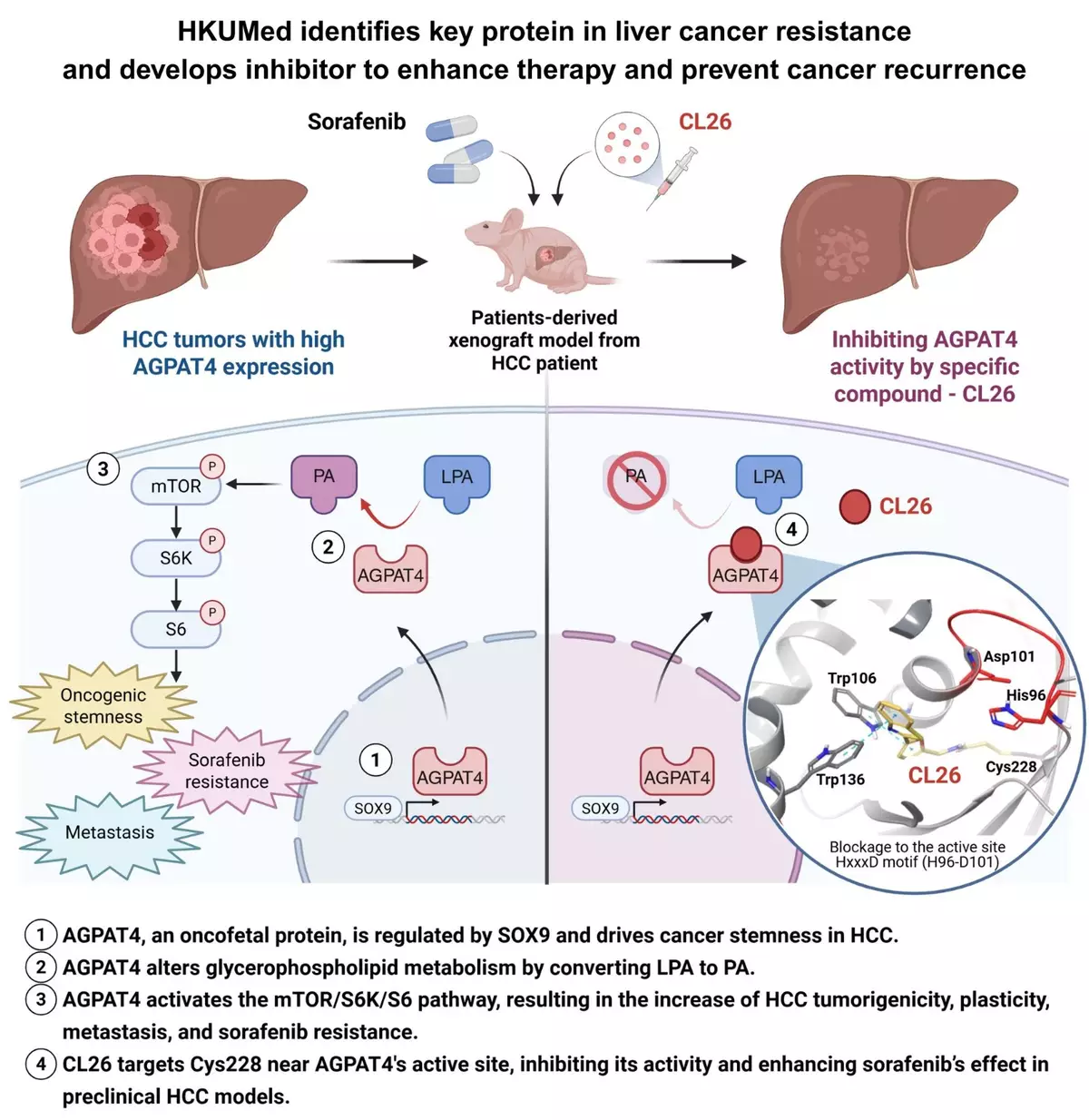
Researchers from the School of Biomedical Sciences at the LKS Faculty of Medicine, the University of Hong Kong (HKUMed), have made a significant advancement that could reshape the treatment landscape for hepatocellular carcinoma (HCC), the most common type of liver cancer, which often resists treatment and recurs. This cancer is especially prevalent in Southeast Asia and China. The research, published in the journal Science Translational Medicine [link to publication], revealed a previously unknown mechanism that allows HCC to grow more aggressively and evade existing therapies. The team also developed a new small molecule inhibitor that could improve treatment options for HCC patients.

A HKUMed research co-led by Professor Stephanie Ma Kwai-yee (first left) and Professor Clive Chung Yik-sham (first right), paves the way for significant advancement in hepatocellular carcinoma (HCC) treatment by identifying a key protein that fuels tumour growth and developing an inhibitor that can improve treatment outcomes.
Unravelling the mystery of tumour resistance
The liver plays a crucial role in various metabolic functions. In HCC, cancerous cells significantly alter these metabolic processes fuelling tumour growth. ‘In our study, we found that a specific metabolic pathway helps cancer cells maintain their stemness, which refer to their enhanced ability to grow, spread and survive,’ explained Professor Stephanie Ma Kwai-yee, co-lead author of the study, and Jimmy and Emily Tang Professor in Molecular Genetics in the School of Biomedical Sciences at HKUMed. Professor Ma is also Associate Vice-President (Research and Innovation) at HKU.
This pathway involves a protein called AGPAT4, which is abundant in both embryonic stem cells and HCC tumour cells, but is rarely found in normal tissues. ‘Our data show that AGPAT4 acts like a switch that makes cancer cells more flexible and aggressive,’ said Professor Ma. ‘This flexibility, known as tumour plasticity, is linked to a higher chance of the cancer to recur, spread and resist existing treatments like sorafenib, a widely used drug for liver cancer.’
The research team discovered that AGPAT4 increases the conversion of a fat molecule called LPA (lysophosphatidic acid) into another molecule called PA (phosphatidic acid). This process activates a signalling pathway involving cell manager mTOR and other proteins, which promote tumour growth and survival. Professor Ma added, ‘When we blocked AGPAT4 in a mouse model, tumour growth slowed down and the cancer became more sensitive to sorafenib.’

HKUMed researchers have identified key protein in liver cancer resistance and developed inhibitor to enhance therapy and prevent cancer recurrence. The research is co-led by Professor Stephanie Ma Kwai-yee (second right) and Professor Clive Chung Yik-sham (first right).
Developing an inhibitor to target AGPAT4
Building on these findings, the team developed a compound called CL26, which specifically targets AGPAT4. In preclinical models using patient-derived tumours, CL26 worked in combination with sorafenib to significantly suppress tumour growth. Toxicological assessments showed minimal side effects, suggesting the compound may be safe for future clinical use.
‘This is a promising step forward,’ said Professor Clive Chung Yik-sham, Assistant Professor in the School of Biomedical Sciences, HKUMed, who also co-led the study. ‘CL26 allows us to target liver cancer cells with precision, potentially improving outcomes for patients who no longer respond to standard treatments.’
Eyes on clinical trials to validate treatment potential
‘Our research suggests that AGPAT4 helps HCC cancer cells become more adaptable and resistant to therapy,’ said Professor Ma. ‘By targeting this protein, we may enhance the effectiveness of sorafenib and expand the therapeutic options for patients in clinical settings. We aim to offer hope to countless patients battling this challenging disease.’Professor Chung added, ‘CL26’s ability to precisely target AGPAT4 is likely one of the key reasons it works so well. We are conducting larger-scale preclinical studies to further assess the efficacy and safety of CL26 and hope to advance to investigational new drug studies and clinical trials.’
About the research team
The collaborative study was co-led by Professor Stephanie Ma Kwai-yee and Professor Clive Chung Yik-sham from the School of Biomedical Sciences at HKUMed. The co-first authors were Dr Johnson Ng Kai-yu and Amy Koo Tin-yan. Major collaborators included professors from The Hong Kong Polytechnic University, the Fourth Military Medical University in Xi’an, the Sun Yat-sen University Cancer Centre, and Guangzhou Medical University in Guangzhou; and Professor Guan Xinyuan from the Department of Clinical Oncology of the School of Clinical Medicine at HKUMed.
Acknowledgements
This research was funded by various grants from the Hong Kong Research Grants Council, including the Theme-Based Research Scheme, the Research Impact Fund, the Collaborative Research Fund, the General Research Fund, the Early Career Scheme and the Research Fellow Scheme. Additional support was provided by the Croucher Foundation’s Senior Research Fellowship and the Guangdong Science and Technology Department. The project also received separate grants through the Innovation and Technology Support Programme under the Innovation and Technology Fund, as well as funding for the Laboratory for Synthetic Chemistry and Chemical Biology, the Centre for Oncology and Immunology, and the Centre for Translational and Stem Cell Biology under the Health@InnoHK Programme initiated by the Innovation and Technology Commission of the HKSAR Government
WASHINGTON (AP) — Deciding when to get routine mammograms is confusing. Some health groups recommend women begin at age 40 or 45 while another recently opted for age 50. They also differ on whether yearly or every other year is best.
The conflicting advice is at least partly because guidelines for breast cancer screening are designed for women at average risk and with no possible cancer symptoms. But breast cancer is so common that it is hard to know who is really “average” and how to balance the pros and cons of screening.
"Breast cancer is not one disease,” said Dr. Laura Esserman of the University of California, San Francisco. “So how in the world does it make sense to screen everybody the same when everyone doesn't have the same risk?”
Esserman is leading research to better understand the nuances of who is at low or high risk or somewhere in between and eventually offer more tailored screening advice.
More than 320,000 women in the U.S. will be diagnosed with breast cancer this year, according to the American Cancer Society. Death rates have been dropping for decades, thanks largely to better treatments. But it is still the second-most common cause of cancer death in U.S. women -- and diagnoses are inching up.
For now, here are some things to know.
The newest guidance comes from the American College of Physicians, which recommends that average-risk women ages 50 to 74 get an every-other-year mammogram. For those 40 to 49, the guideline says to discuss pros and cons with a doctor and if they choose screening, to go every other year.
That advice, issued last month, was a surprise. Most other U.S. health groups have urged women to start earlier, in their 40s. The influential U.S. Preventive Services Task Force recently switched its guidance to start every-other-year mammograms at age 40 instead of 50.
The American Cancer Society has long recommended yearly mammograms for 45- to 54-year-olds -– but says they can choose to start at 40. For those age 55 and older, the cancer society says women can switch to every other year or choose to keep going for yearly checks.
The new American College of Physicians guidelines also say doctors can ask if women 75 or older wish to stop routine screening. In contrast, the cancer society says there is no reason to stop if they are still healthy.
The higher a woman’s risk of eventually developing breast cancer, the more benefit she will derive from more frequent screenings. But beyond some well-known factors like the cancer-causing BRCA1 or BRCA2 genes, it is hard for women to know their true risk. Age has long been a proxy because the risk of breast cancer rises as women get older.
Mammograms aren’t perfect. Sometimes they miss cancer or an aggressive tumor pops up after a routine mammogram. But guidelines seek to balance the benefits of catching cancer early with possible harms, such as stress and pain from investigating suspicious spots that don't turn out to be cancerous.
“We’re not saying there’s no benefit” from mammograms in the 40s, cautioned Dr. Carolyn Crandall of the University of California, Los Angeles, who chaired the American College of Physicians report. But “there’s a narrower balance between the benefits you could get and the harms in 40- to 49-year-olds.”
The American Cancer Society recommends starting yearly mammograms at 45 because it found breast cancer incidence in 45- to 49-year-olds was higher than in the early 40s – more like what 50- to 54-year-olds experience, said public health researcher Robert Smith, the society’s expert on early cancer detection.
What is missing is a way to tell if someone is more likely to develop an aggressive breast cancer or a slow-growing one, Smith noted.
Nearly half of women over 40 have dense breast tissue, which can make it harder to spot a tumor on a mammogram and can slightly increase the risk of developing cancer.
After a mammogram, women are notified about their breast density. Many experts say it is not yet clear if women with dense breasts would benefit from adding ultrasounds or MRIs to their screening. But the new American College of Physicians guidance advises considering 3D mammography – what doctors call digital breast tomosynthesis or DBT.
In the future, adding a gene test — one that looks at more than just those well-known BRCA genes — along with broader risk factors may help refine women’s optimal mammogram schedule.
A recent study of nearly 46,000 women, called the WISDOM trial, used age, genetic testing, lifestyle, health history and breast density to classify women as low, average, elevated or high risk. That risk level determined if they waited to start mammograms at 50, went every other year or every year – and the highest-risk group was told to screen twice a year, once with a mammogram and again with an MRI scan. Risk-based scans were compared to standard yearly mammograms.
Risk-based screening worked as well as yearly screening, Esserman's team reported in the medical journal JAMA. One surprise: About 30% of women whose gene testing indicated increased risk didn't report relatives with breast cancer. While more research is underway, Esserman hopes the early findings will start influencing guidelines soon.
Also in the pipeline are AI tools being crafted to assess a woman’s risk of developing breast cancer in the next few years based on clues in her mammogram, another possible way to identify who might qualify for more or less frequent screening.
For now, women can talk with their doctors about close relatives who have had cancer, their own overall health and other risk factors such as whether they have had children and at what age.
Whatever mammogram age and interval they choose, the best advice is to stick with it, the cancer society's Smith said: “Breast screening works best when it’s done regularly.”
The Associated Press Health and Science Department receives support from the Howard Hughes Medical Institute’s Department of Science Education and the Robert Wood Johnson Foundation. The AP is solely responsible for all content.
FILE - A radiologist uses a magnifying glass to examine mammograms for breast cancer in Los Angeles, May 6, 2010. (AP Photo/Damian Dovarganes, File)













