Flu activity has surged in many parts of the Northern Hemisphere, driven primarily by a newly emerged H3N2 strain known as 'subclade K'. A research team led by the Department of Microbiology and the Department of Medicine, both under the School of Clinical Medicine at the LKS Faculty of Medicine of the University of Hong Kong (HKUMed), has found that most hospital patients in Hong Kong have little to undetectable levels of neutralising antibodies against this mutated strain. HKUMed researchers strongly advise the public to get vaccinated as soon as possible.

HKUMed study reveals low immunity against H3N2 strain in Hong Kong and urges early vaccination.
Neutralising antibody levels are closely linked to protection against infection. The research team was led by Professor Kelvin To Kai-wang, Clinical Professor and Chairperson of the Department of Microbiology, and Professor Ivan Hung Fan-ngai, Chair Professor and Head of the Division of Infectious Diseases in the Department of Medicine of the School of Clinical Medicine at HKUMed. The team collected serum specimens from public hospitals to monitor viral genetics and antibody responses and assess the overall immunity level against H3N2 in Hong Kong.
Analysis of 277 serum samples collected in November 2025 revealed that 52% of individuals had detectable neutralising antibodies against the previously circulating subclade J.2.2, with 27% reaching a titer of 40 or above, indicating a relatively high level of immunity. In stark contrast, only 18% of individuals had detectable antibodies against the newly emerged subclade K, with 0.7% reaching a titer of 40 or above.
'Our findings suggest that the local community's immune barrier against the subclade K is insufficient and lower than that against the previously circulating subclade J.2.2,' explained Professor Ivan Hung.

Professor Ivan Hung Fan-ngai (left) said the findings suggest that the local community’s immune barrier against the subclade K is insufficient and lower than that against the previously circulating subclade J.2.2.
To reduce the risk of severe infection, the research team recommends the following measures:
• Get vaccinated against influenza. A study from England found that vaccine effectiveness against emergency department visits and hospital admissions is between 72% and 75% for children and adolescents, and 32% to 39% for adults.
• Seek early diagnosis using rapid antigen tests.
• Consult a doctor promptly. Antiviral medications are most effective when administered within 48 hours of symptom onset.
Professor Kelvin To emphasised, 'These data serve as a clear alert for Hong Kong. With influenza A cases rising and new variants emerging, we must reinforce the message that vaccination is the most effective way to prevent infection. For those who do become infected, seeking medical care promptly is essential to reduce the risk of severe complications.'
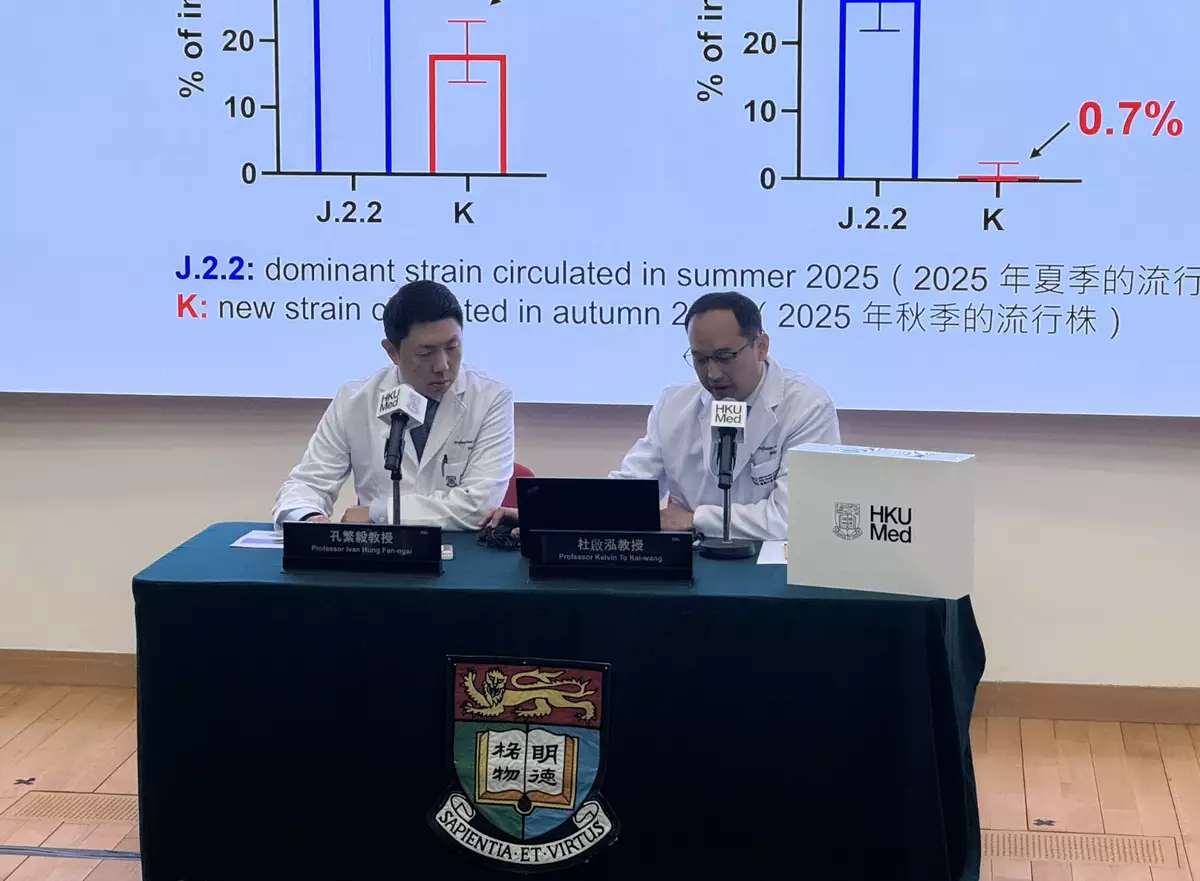
Professor Kelvin To Kai-wang (right) emphasised that for those who become infected, seeking medical care promptly is essential to reduce the risk of severe complications.
WASHINGTON (AP) — Defense Secretary Pete Hegseth announced Tuesday that the U.S. military will no longer require all American troops to get the flu vaccine, citing “medical autonomy” and religious freedom.
“The notion that a flu vaccine must be mandatory for every service member, everywhere, in every circumstance at all times is just overly broad and not rational,” Hegseth said in a video posted on social media.
He said American service members are free to get the flu vaccine but will not be forced to “because your body, your faith and your convictions are not negotiable.”
Hegseth’s directive does allow for the military services to request to keep the vaccine requirement in place, according to a memo enacting the policy posted online. It says the services have 15 days to make those requests.
Vaccination programs in the U.S. military date back to the American Revolution. But they became a contentious political issue during the coronavirus pandemic, when more than 8,400 troops were forced out of the military for refusing to obey the 2021 mandate for the COVID-19 vaccine. Thousands of others sought religious and medical exemptions.
Congress agreed to rescind the mandate, which the Pentagon dropped in January 2023, after roughly 99% of active duty troops in the Navy, Air Force and Marine Corps had gotten the vaccine, and 98% of those in the Army. The Guard and Reserve rates are lower but generally are more than 90%.
The Trump administration then spent months crafting a policy to allow service members who refused to take the mandatory COVID-19 vaccine to reenter service with back pay. While only a tiny fraction have taken the Pentagon up on the new policy, Hegseth’s team has spent the past several months personally highlighting them.
The Pentagon stated in March that 153 service members who were separated under the COVID-19 mandate had been reinstated or "re-accessed."
The dropping of the flu vaccine mandate follows what health officials said was a particularly severe flu season when U.S. infections surged. Public health experts recommend that everyone 6 months and older get an annual influenza vaccine.
The Trump administration has been working to dial back vaccine recommendations. It stated earlier this year that it will no longer recommend flu shots and some other types of vaccines for all children, saying it’s a decision parents and patients should make in consultation with their doctors. A federal judge has temporarily blocked that effort as a lawsuit plays out.
The Congressional Research Service listed eight mandatory vaccines for service members in a 2021 report. They included vaccines for the flu, polio and tetanus as well as the measles and hepatitis A and B.
Service members could request to opt out of a vaccine requirement for religious reasons, the report stated. But the unit commander was required to seek input from medical and religious representatives, while also counseling the service member on the potential impact on their ability to deploy. A military physician also had to counsel the service member on the benefits and risks of forgoing a required vaccination.
The Congressional Research Service noted that the military instituted its first vaccination program in 1777 when Gen. George Washington directed the inoculation of the Continental Army to protect personnel from smallpox.

Defense Secretary Pete Hegseth speaks to members of the media during a press briefing at the Pentagon, Thursday, April 16, 2026 in Washington. (AP Photo/Kevin Wolf)