WALNUT CREEK, Calif.--(BUSINESS WIRE)--Jan 26, 2026--
Central Garden & Pet Company (NASDAQ: CENT) (NASDAQ: CENTA) (“Central”), a leading consumer goods company in the pet and garden industries, today announced the appointment of Apur Patel as Chief Legal Officer, effective January 26, 2026. Mr. Patel succeeds George Yuhas, who will retire from the company.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260126694850/en/
In this role, Mr. Patel will oversee all legal matters for Central, including corporate governance, mergers and acquisitions, securities and compliance, litigation, intellectual property, and enterprise risk management. He will report directly to Niko Lahanas, Chief Executive Officer of Central Garden & Pet.
“We’re grateful to George for stepping back into the role on an interim basis and helping ensure a smooth transition,” said Niko Lahanas. “As George returns to retirement, we’re excited to welcome Apur to Central. Apur brings deep experience and a strong track record of aligning legal strategy with business priorities while effectively managing risk in complex, global organizations.”
Mr. Patel is a senior in-house legal executive with more than 30 years of global experience across consumer packaged goods, technology, and healthcare. He joins Central from The Clorox Company, where he most recently served as Vice President & Deputy General Counsel, leading a team of more than 20 attorneys and paralegals and overseeing Intellectual Property, Commercial and Technology Transactions, Licensing, and Marketing Communications and Advertising.
Earlier in his career, Mr. Patel held senior legal roles at Cisco, Cargill, and UnitedHealth Group, supporting complex global businesses and leading cross-functional initiatives. He is widely respected for his executive presence, strong business acumen, and ability to build and lead high-performing legal teams.
“I am excited to join Central Garden & Pet,” said Mr. Patel. “Central’s entrepreneurial culture, strong portfolio of leading brands, and commitment to long-term value creation are compelling. I look forward to building on the strong legal foundation established by George and partnering with Niko and the leadership team to support Central’s continued success.”
Mr. Patel holds a Juris Doctor degree from Washington and Lee University School of Law and a bachelor’s degree from Colgate University.
About Central Garden & Pet
Central Garden & Pet Company (NASDAQ: CENT) (NASDAQ: CENTA) is a leading consumer goods company in the pet and garden industries. Guided by the belief that home is central to life, the Company's purpose is to proudly nurture happy and healthy homes. For over 45 years, its innovative and trusted solutions have helped lawns grow greener, gardens bloom bigger, pets live healthier, and communities grow stronger.
Central is home to a diversified portfolio of market-leading brands including Amdro ®, Aqueon ®, Best Bully Sticks ®, Cadet ®, C&S ®, Farnam ®, Ferry-Morse ®, Kaytee ®, Nylabone ®, Pennington ®, Sevin ® and Zoёcon ®. With fiscal 2025 net sales of $3.1 billion, Central has strong manufacturing and logistics capabilities supported by a passionate, entrepreneurial growth culture. The Company is headquartered in Walnut Creek, California, and employs over 6,000 people, primarily across North America. Visit www.central.com to learn more.

Apur Patel joins Central Garden & Pet as Chief Legal Officer, overseeing all legal matters for the company, including Corporate Governance, effective January 26, 2026.
CHARLOTTE, N.C.--(BUSINESS WIRE)--Apr 7, 2026--
Kerecis, the company pioneering the use of sustainably sourced fish-skin in cellular therapy and tissue regeneration, will showcase its intact fish-skin technology and highlight new clinical data on pressure ulcers at the Symposium on Advanced Wound Care (SAWC) Spring 2026, taking place April 8-11 at the Charlotte Convention Center in Charlotte, North Carolina. The company will also emphasize expanded multi-specialty adoption. Visitors can explore Kerecis' portfolio of intact fish-skin grafts and complementary Biatain wound dressing solutions at Booth 1508, clinical insights and clinical trial results on the intact fish-skin platform will be presented at an industry-supported symposium, while clinicians present 13 abstracts on intact fish-skin technology during the symposium.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260407701398/en/
The industry-supported symposium titled, “Reeling in Results: Clinical Applications of Fish-Skin Grafts in Chronic and Atypical Wounds.” will take place on Thursday, April 9, 2026, from 4:30 PM to 5:30 PM EDT, and will feature clinical insights on real-world applications of fish-skin grafts across complex and atypical wounds and the new clinical research.
The clinical insights on real-world applications of fish-skin grafts across complex and atypical wounds will be presented by Dr. Anamika Agrawal, Dr. John C. Lantis II, and Carrie McGroarty, PA-C, MPAS, EdD.
The new clinical research will be presented by Dr. John C. Lantis II. The real-world trial found that intact fish-skin demonstrated superior clinical effectiveness when compared with standard care for treating severe, hard-to-heal Stage 3 and 4 pressure ulcers in 80 patients.
Key findings include:
The research was authored by Dr. Hongyu Miao (Florida State University), Ben LeBoutillier (Intellicure, LLC), Dr. John C. Lantis II (Mount Sinai West and Icahn School of Medicine), and Dr. Caroline Fife (Intellicure, LLC; Baylor College of Medicine). The study evaluated a highly complex, real-world patient population with only 37.5% of the patients being ambulatory and a high prevalence of severe comorbidities, including diabetes (27.5%), peripheral artery disease (25.0%), and autoimmune disease (25.0%) and is available as a pre-print.
"These findings provide crucial, real-world validation for the clinical and economic value of the Kerecis intact fish-skin technology," said Fertram Sigurjonsson, Coloplast Executive Vice President and Kerecis Inventor & Founder. "Stage 3 and 4 pressure ulcers represent a massive burden to patients and the broader healthcare system. Demonstrating statistical superiority in this highly complex cohort underscores the vital medical necessity of advanced biologic therapies when standard, conservative measures stall."
Furthermore, 13 abstracts spanning multiple specialties, underscoring the growing body of evidence supporting intact fish-skin technology will be presented at the symposium’s poster reception on Friday, April 10, 2026, 7:30 PM - 8:30 PM EDT in Hall C1.
With participation from surgeons, podiatrists, vascular specialists, and advanced practice providers, Kerecis’ presence at SAWC Spring reflects continued adoption across specialties and care settings, supported by expanding clinical evidence and real-world experience.
Visitors to Booth 1508 will have the opportunity to engage with clinical experts, explore new data, and learn how intact fish-skin technology is being applied to address real-world challenges in tissue repair.
About Kerecis
Kerecis, founded by Fertram Sigurjonsson, develops intact fish-tissue derived products for cellular therapy, tissue regeneration, and protection. When grafted onto damaged human tissue or implanted, the patented material supports the body’s own processes to heal and regenerate. Because no viral or prion transfer risk exists between Atlantic cod and humans, Kerecis products are only gently processed and retain their similarity to human tissue. The gentle processing preserves the material’s original three-dimensional structure, maintaining its inherent natural strength, complexity, and molecules (such as fatty acids). Clinical studies show that Kerecis products heal wounds faster than competitors. Kerecis is the only global manufacturer of medical devices containing intact fish-tissue and is the fastest growing company in the U.S. xenograft biologics skin market. Products include SurgiBind ® /SurgiClose ®, GraftGuide ®, MariGen ®, and Shield™ for various medical applications. Committed to the UN Sustainable Development Goals, Kerecis uses sustainably sourced Icelandic fish processed with renewable energy. Kerecis is a part of Coloplast, a leading supplier of intimate healthcare products. For more information about Kerecis and its clinical research, visit www.kerecis.com.
Trademarks and registered trademarks are the property of their respective owners.
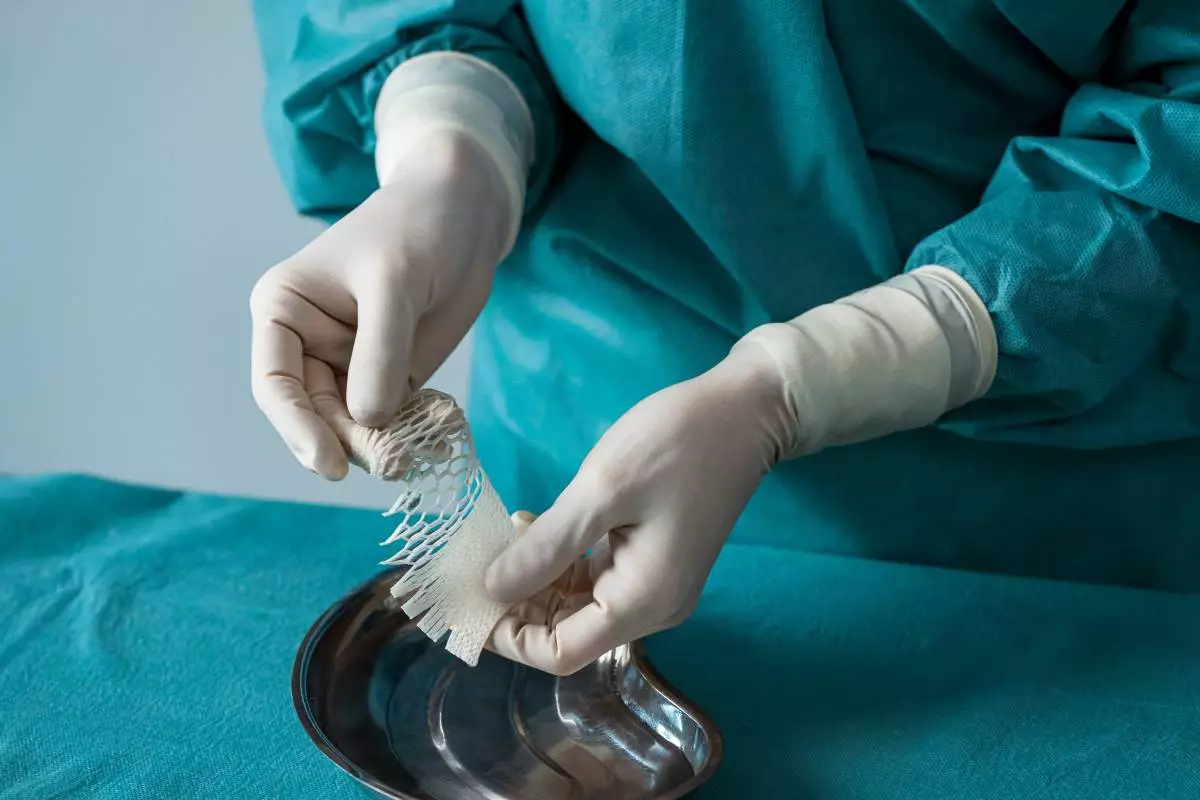
Kerecis mesh Clinic Gx large

Steve Dunn, Kerecis Exhibitor at Symposium on Advanced Wound Care (SAWC) Spring 2026, April 8-11












