|
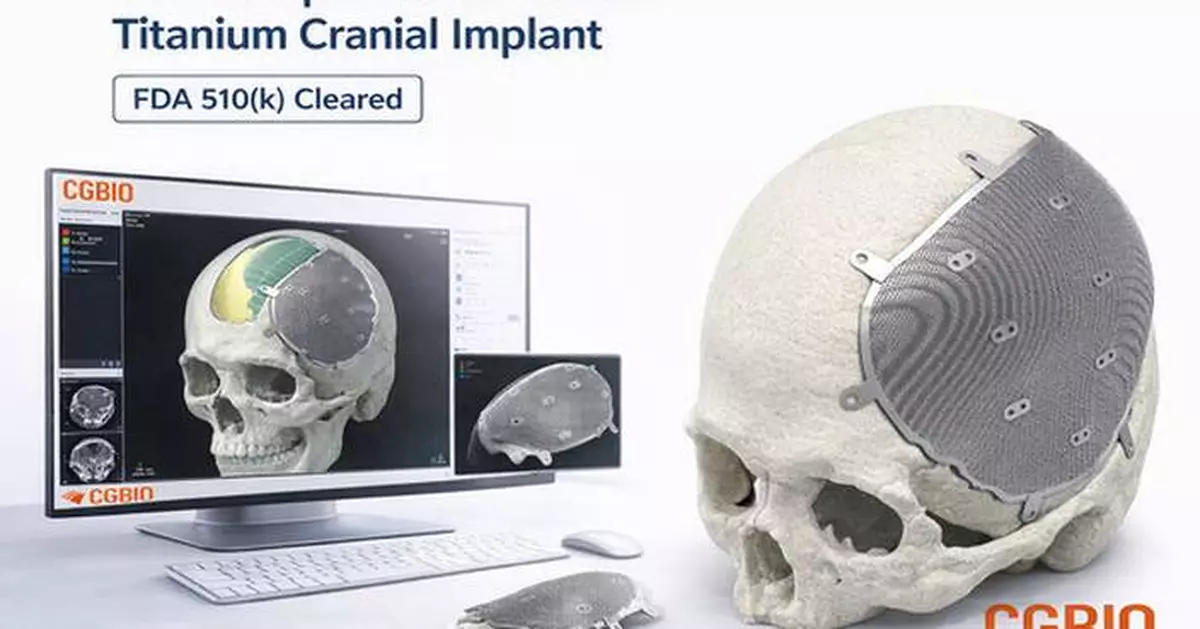
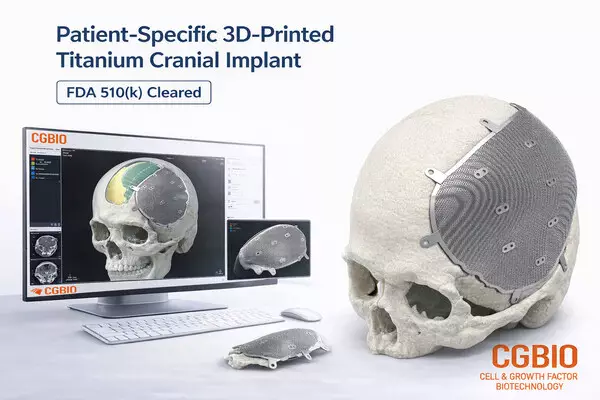
- Patient-specific titanium implant designed for cranial and craniofacial reconstruction
- First Korean company to obtain FDA 510(k) clearance for a customized titanium implant in the U.S. market
SEOUL, South Korea, April 24, 2026 /PRNewswire/ -- CGBIO (CEO: Hyunseung Yu) announced that its patient-specific titanium implant, EASYMADE TI, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on April 9. The FDA 510(k) pathway is a premarket clearance process that demonstrates substantial equivalence to a legally marketed predicate device, allowing commercialization in the United States.
This milestone marks the first case of a Korean company obtaining FDA 510(k) clearance for a patient-specific titanium implant intended for the U.S. market. It highlights CGBIO's technological capabilities as well as its regulatory expertise in global markets. The achievement is particularly notable in the field of patient-specific cranial and craniofacial reconstruction.
EASYMADE TI is a patient-specific titanium implant designed based on individual CT data and intended for reconstruction of cranial and non-load-bearing craniofacial defects. The implant is manufactured using medical-grade titanium alloy and produced through Laser Powder Bed Fusion (LPBF), an advanced metal 3D printing technology.
The product delivery process is also a key differentiator. Once patient CT images are provided by U.S.-based clinicians, customized design is completed at CGBIO's design center in Korea, and the finished implant is delivered to hospitals in the U.S. within five days. After sterilization at the hospital, the implant can be used directly in surgical procedures.
The company expects this clearance to serve as a turning point for expanding its patient-specific implant business in the United States. Demand for cranial and craniofacial reconstruction continues to grow in the U.S., driven by cases such as trauma, tumor resection, and decompressive craniectomy. At the same time, interest in patient-specific reconstruction solutions tailored to individual anatomy is increasing.
Hyunseung Yu, CEO of CGBIO, said, "The FDA 510(k) clearance of EASYMADE TI is a meaningful achievement that demonstrates CGBIO's global competitiveness in patient-specific reconstruction solutions. We will use this milestone as a foundation to expand our customized cranial and craniofacial implant business in the U.S. and accelerate regulatory approvals and commercialization of our pipeline products."
CGBIO plans to actively expand its presence in the U.S. market based on this clearance, while continuing to strengthen its global position in the field of patient-specific medical devices.
- Patient-specific titanium implant designed for cranial and craniofacial reconstruction
- First Korean company to obtain FDA 510(k) clearance for a customized titanium implant in the U.S. market
SEOUL, South Korea, April 24, 2026 /PRNewswire/ -- CGBIO (CEO: Hyunseung Yu) announced that its patient-specific titanium implant, EASYMADE TI, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on April 9. The FDA 510(k) pathway is a premarket clearance process that demonstrates substantial equivalence to a legally marketed predicate device, allowing commercialization in the United States.
This milestone marks the first case of a Korean company obtaining FDA 510(k) clearance for a patient-specific titanium implant intended for the U.S. market. It highlights CGBIO's technological capabilities as well as its regulatory expertise in global markets. The achievement is particularly notable in the field of patient-specific cranial and craniofacial reconstruction.
EASYMADE TI is a patient-specific titanium implant designed based on individual CT data and intended for reconstruction of cranial and non-load-bearing craniofacial defects. The implant is manufactured using medical-grade titanium alloy and produced through Laser Powder Bed Fusion (LPBF), an advanced metal 3D printing technology.
The product delivery process is also a key differentiator. Once patient CT images are provided by U.S.-based clinicians, customized design is completed at CGBIO's design center in Korea, and the finished implant is delivered to hospitals in the U.S. within five days. After sterilization at the hospital, the implant can be used directly in surgical procedures.
The company expects this clearance to serve as a turning point for expanding its patient-specific implant business in the United States. Demand for cranial and craniofacial reconstruction continues to grow in the U.S., driven by cases such as trauma, tumor resection, and decompressive craniectomy. At the same time, interest in patient-specific reconstruction solutions tailored to individual anatomy is increasing.
Hyunseung Yu, CEO of CGBIO, said, "The FDA 510(k) clearance of EASYMADE TI is a meaningful achievement that demonstrates CGBIO's global competitiveness in patient-specific reconstruction solutions. We will use this milestone as a foundation to expand our customized cranial and craniofacial implant business in the U.S. and accelerate regulatory approvals and commercialization of our pipeline products."
CGBIO plans to actively expand its presence in the U.S. market based on this clearance, while continuing to strengthen its global position in the field of patient-specific medical devices.
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **
CGBIO Receives FDA 510(k) Clearance for Patient-Specific Titanium Implant 'EASYMADE TI'
WUHU, China, April 24, 2026 /PRNewswire/ -- Beijing Auto Show 2026 opened on April 24, bringing together global automotive brands and advanced technologies. As a key barometer of the industry, the show serves as a platform for innovation and exchange. Chery Group appeared with its full brand lineup, including CHERY, EXEED, iCAUR, OMODA & JAECOO, and LEPAS. A total of 15 models made their global debut. The event welcomed over 4,000 guests from more than 100 countries, setting a new record for Chery in both scale and international reach.
At the show, Chery presented breakthrough technologies and a forward-looking strategy. Built on a global brand ecosystem, core technology development, and new energy innovation, the company demonstrated capabilities reshaping industry competition and setting new benchmarks for value creation.
Deepening Global Layout: A Multi-Brand Portfolio Reshaping the Industry Landscape
With a five-brand portfolio developed through years of overseas experience, Chery has formed a multi-scenario ecosystem. Each brand targets specific segments, shifting from broad coverage to precise positioning. Key debuts included Tiggo V, ES GT, EX8, OMODA 4, right-hand-drive V27, and L6 BEV, covering family, premium, and youth mobility needs. The brands operate in synergy, sharing resources and technologies to drive Chery's shift from scale expansion to value upgrading.
Advanced Architecture Redefines the Upper Limit of Experience: Digital Chassis Breaks the Boundaries of Smart Mobility
Chery showcased next-generation architecture innovations. The Feiyu Digital Intelligent Chassis i integrates steer-by-wire and brake-by-wire, enabling 90 km/h moose test performance and zero-radius tank turns. The GAIA All-Domain System combines amphibious mobility with satellite communication, redefining the vehicle as an all-scenario mobility terminal. The industry-first full-domain 48V system improves efficiency by 15% and meets ASIL D safety standards.
Through continuous breakthroughs, Chery is evolving from a technology follower into a standards setter in global smart mobility.
Full-Chain Energy Efficiency Management Enables Technology Accessibility: System Innovation Addressing Global Mobility Challenges
Chery introduced a full life-cycle energy efficiency system. The KunPeng High Efficiency Engine achieves 48.57% thermal efficiency and 4.0 kWh/L conversion. The DHT360 hybrid system delivers up to 360 kW, enabling full-speed electric driving. The Rhino Battery offers high safety and fast charging, reaching 80% in 12 minutes. The system supports a more efficient and sustainable mobility future.
Website: https://www.cheryinternational.com
** This press release is distributed by PR Newswire through automated distribution system, for which the client assumes full responsibility. **

Driving the Future of Mobility with Technology: Chery at Beijing Auto Show 2026