BOSTON--(BUSINESS WIRE)--Apr 2, 2026--
Seaport Therapeutics (“Seaport” or the “Company”), a clinical-stage therapeutics company advancing novel neuropsychiatric medicines with a proven strategy and team, today announced positive topline data from its single-ascending dose (SAD) and crossover portions of its Phase 1 proof-of-concept clinical trial evaluating GlyphAgo™ (SPT-320™ or Glyph Agomelatine), a novel, Glyphed oral prodrug of agomelatine in development for generalized anxiety disorder (GAD). The clinical proof-of-concept topline results demonstrated that GlyphAgo exceeded the program’s target of a 2-fold increase in bioavailability compared to unmodified agomelatine, achieving therapeutic levels of agomelatine at substantially lower doses that reduce liver exposure and are projected to reduce or eliminate the need for liver function testing.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260402742148/en/
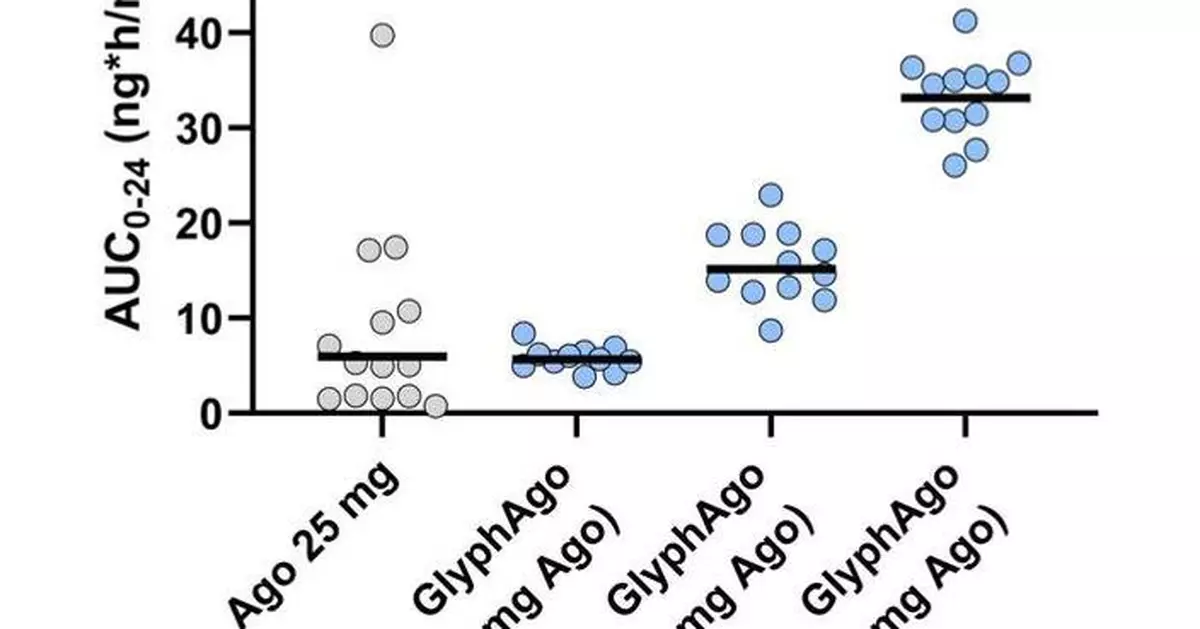
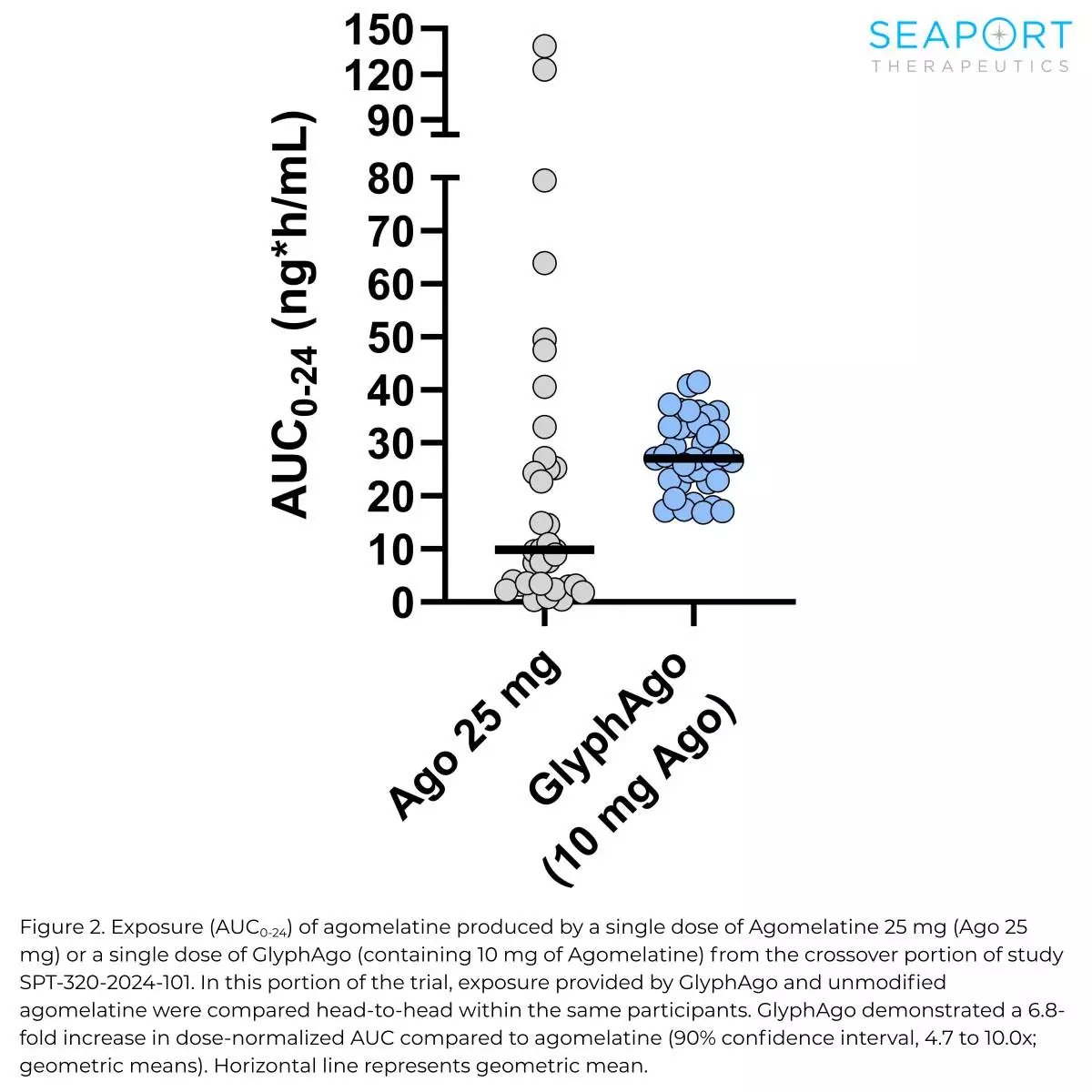
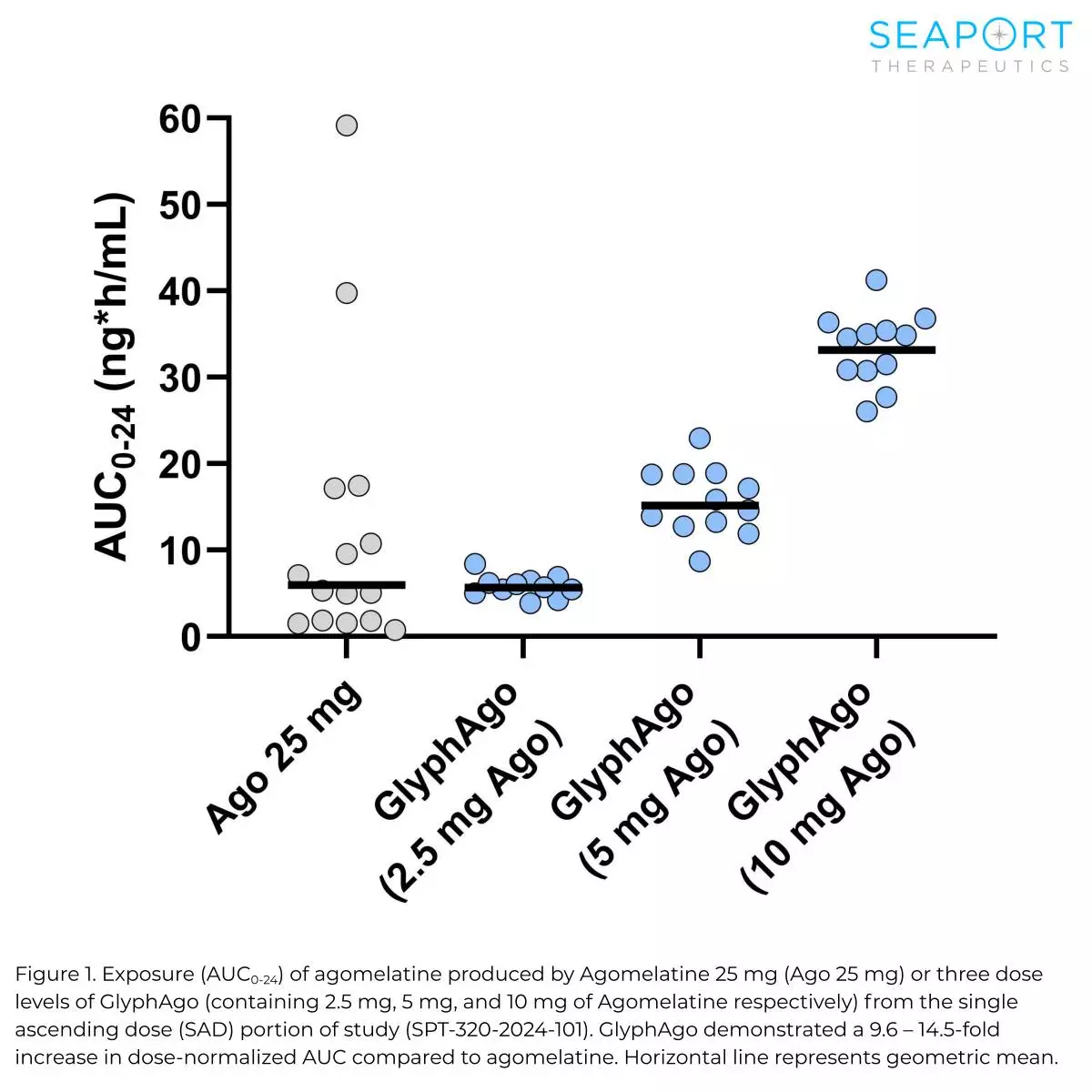
In the head-to-head crossover portion of the trial, GlyphAgo demonstrated a 6.8-fold increase in bioavailability of agomelatine compared to unmodified orally administered agomelatine. GlyphAgo also showed significantly lower (10-fold) PK variability compared to unmodified agomelatine. The crossover portion included participants who were taking estrogen-containing oral contraceptives that are known to increase agomelatine exposure due to liver drug-drug interaction. In contrast, GlyphAgo exposure was unaffected by oral contraceptives, further supporting the ability of GlyphAgo to bypass first-pass liver metabolism. GlyphAgo demonstrated a 9.6 to 14.5-fold increase in dose-normalized exposure compared to agomelatine in a separate SAD portion of the trial in which no participants were on oral contraceptives. GlyphAgo was well tolerated and no liver-related adverse events (AEs) were observed. The completed SAD and crossover portions of the trial, which included approximately 130 participants, conclude the PK proof-of-concept objectives of the trial, while the ongoing multiple-ascending dose (MAD) portion of this trial – conducted with only GlyphAgo – is intended to further characterize the safety and PK of repeat-dosing of GlyphAgo.
“These topline data, from a well-powered Phase 1 trial, strengthen our conviction in GlyphAgo’s potential and provide further clinical validation for the Glyph platform,” said Daphne Zohar, Co-Founder and Chief Executive Officer at Seaport Therapeutics. “Based on these data, we plan to advance GlyphAgo into two parallel trials, a Phase 2a proof-of-pharmacology trial to evaluate the potential sleep benefit of GlyphAgo in patients with GAD, and a Phase 2b trial in GAD, that is a randomized placebo-controlled trial designed to be registration-enabling. We believe that GlyphAgo has the potential to bring patients with generalized anxiety disorder what could be the first new therapy in decades in the U.S. for this underserved and debilitating disorder.”
Agomelatine, a clinically validated MT1/MT2 melatonin receptor agonist and serotonin 2C (5-HT2C) receptor antagonist, is an effective anxiolytic and antidepressant approved for the treatment of GAD in Australia and major depressive disorder (MDD) in Australia and the European Union (EU). Agomelatine’s label in both Australia and the EU requires liver function testing before initiating treatment, during treatment, and upon increasing the dose. Agomelatine is not approved in the U.S.
“In GAD, agomelatine has demonstrated robust and statistically significant separation from placebo in four third-party placebo-controlled studies 1-4 and has been observed in meta-analysis to have better efficacy and tolerability than selective serotonin-reuptake inhibitors or benzodiazepines,” said Steven Paul, M.D., Co-Founder and Board Chair at Seaport Therapeutics. “Despite this positive profile, over 90 percent of unmodified agomelatine is lost to first-pass metabolism and its use has been limited by dose-dependent liver enzyme elevations. The enhanced pharmaceutical properties of GlyphAgo and resulting markedly reduced inter-individual variability in systemic exposure to agomelatine support our clinical development of GlyphAgo in GAD.”
Using Seaport’s proprietary Glyph™ platform, GlyphAgo is designed to enhance lymphatic absorption and avoid first-pass liver metabolism, thereby enhancing oral bioavailability and reducing side effects. By leveraging an alternative absorption pathway via the intestinal lymphatic system used by dietary fats, GlyphAgo is designed to increase systemic exposure of agomelatine, enabling exposure levels of agomelatine that are effective in GAD but at a lower dose that reduces liver exposure and reduces or eliminates the need for liver function testing. Based on the data that Seaport has generated to date, GlyphAgo has the potential to become a leading treatment for GAD.
“Agomelatine combines a differentiated mechanism with a favorable efficacy and tolerability profile in GAD, but its potential has been previously limited by first-pass liver metabolism and the need for burdensome liver testing,” said Daniel Bonner, Ph.D., Co-Founder and Senior Vice President, Platform, at Seaport Therapeutics. “These results show that GlyphAgo exceeded the targeted improvement in bioavailability, achieving robust exposure, and a more consistent PK profile at a substantially lower dose of agomelatine.”
Phase 1 Trial Design
The Phase 1 proof-of-concept trial is being conducted in multiple parts to evaluate the safety, tolerability, and PK of GlyphAgo and to compare the PK of GlyphAgo to agomelatine alone. The trial includes single and multiple-ascending dose cohorts, as well as a crossover portion, (including both food-effect and within-participant comparison between GlyphAgo and agomelatine), using both open-label and placebo-controlled designs. In the SAD portion of the Phase 1 trial, healthy volunteers received a single administration of either ascending doses of GlyphAgo or a 25 mg dose of agomelatine, an approved efficacious dose in Australia and the EU, to assess PK parameters, including area under the curve (AUC), a measure of overall exposure, and Cmax, or peak plasma concentrations. In the SAD portion, participants were healthy volunteers with no evidence of liver impairment who were not taking any medications or supplements known to alter the PK of agomelatine, including fluvoxamine or estrogen-containing oral contraceptives. In the crossover portion, participants were randomized to one of two sequences designed to assess the food effect on a single dose level of GlyphAgo and compare it with a 25 mg dose of agomelatine. Both sequences evaluated GlyphAgo under fed and fasted conditions before agomelatine, a structure chosen to avoid confounding GlyphAgo safety results with agomelatine’s known liver toxicity.
Topline Results
Crossover Portion
SAD Portion
Seaport plans to present additional analyses from the Phase 1 trial, including the results from the MAD portion, at future upcoming scientific meetings.
About GlyphAgo (SPT-320 or Glyph Agomelatine)
GlyphAgo is a novel, Glyphed oral prodrug of agomelatine, a clinically validated anxiolytic and antidepressant that is approved for the treatment of GAD in Australia and MDD in Australia and the EU. Using Seaport’s proprietary Glyph platform, GlyphAgo is designed to avoid first-pass liver metabolism and increase systemic exposure of agomelatine, enabling exposure levels of agomelatine that are effective in GAD but at a lower dose that reduces liver exposure and reduces or eliminates the need for liver function testing. Based on internal analyses, Seaport believes a two-fold increase in the bioavailability of agomelatine with GlyphAgo dosing will reduce or eliminate liver enzyme elevations. Based on the data generated to date, Seaport believes GlyphAgo has the potential to become a leading treatment for GAD.
About Seaport Therapeutics
Seaport Therapeutics is a clinical-stage therapeutics company advancing the development of novel neuropsychiatric medicines in areas of high unmet patient needs. The Company has a proven strategy of advancing clinically validated mechanisms previously held back by limitations that are overcome with its proprietary Glyph technology platform. All the therapeutic candidates in its pipeline of first and best-in-class medicines are based on the Glyph platform, which is uniquely designed to enable oral bioavailability, bypass first-pass metabolism and reduce liver enzyme elevations or hepatotoxicity and other side effects. Seaport is led by an experienced team that invented and advanced important neuropsychiatric medicines and is guided by an extensive network of renowned scientists, clinicians, and key opinion leaders. For more information, please visit www.seaporttx.com.
Seaport Therapeutics Announces Positive Proof of Concept Topline Results from Ongoing Phase 1 Trial of GlyphAgo™ in Healthy Volunteers
Seaport Therapeutics Announces Positive Proof of Concept Topline Results from Ongoing Phase 1 Trial of GlyphAgo™ in Healthy Volunteers