DES MOINES, Iowa (AP) — The near-daily changes in U.S. gas prices are dizzying for drivers, who are left feeling frustrated and cash-strapped by the highest fuel costs since 2022.
With the Iran war pushing up prices worldwide, the U.S. average for a gallon of gas topped $4 on Tuesday, according to AAA.
Prices can change from one day to the next or from one nearby station to another, forcing drivers in the U.S. to game out the right moment to fill up or hunt for cheaper prices.
Experts say differences in price aren’t typically decided by any individual gas retailer, and most of them aren't pocketing the extra pennies when prices rise. The uncertainty at the pump is trickling down from a massive, volatile oil and gas market that's making it hard for gas stations to keep up.
Lonnie McQuirter, director of operations at 36 Lyn Refuel Station in south Minneapolis, said his margins have gotten much tighter. About a mile (1.6 kilometers) off Interstate 35, the neighborhood convenience store posted $3.399 a gallon for regular gas on Wednesday, which is about 18 cents lower than the metro average, according to AAA.
“We price based on what we’re able to buy fuel at, and how well we can operate,” McQuirter said. He declined to speculate about his competitors, saying, “They’ve got different economics.”
Wholesale fuel prices, which shift multiple times a day, are the main reason McQuirter said he's charging more than a month ago. He's also facing higher credit card fees and rising costs to maintain pumps.
In times like these, with consumers “screaming for help,” McQuirter said small operators like him act more on emotion than greed.
“We’re in our stores every day looking our customers in the eye,” he said. “It really takes a toll when people are having to cut back on certain things in order to afford to live.”
A lot of it is outside the gas retailer’s control. Roughly half the price at the pump pays for the cost of crude oil, the main ingredient in gasoline, according to the U.S. Energy Information Administration. About 20% goes to refiners who turn crude into gas.
Those costs have risen as crude oil prices jumped in response to the war and shipping disruptions in the Strait of Hormuz. Gas retailers are adjusting the price at the pump to account for the higher price they just paid for their next shipment of gasoline.
Taxes — federal, state and local — account for nearly 20% of the price, while about 10% is left for retailers, who still have to pay for transportation, labor and other expenses.
Retailers' markup has averaged about 38 cents a gallon over the past five years, according to the convenience store trade group NACS, citing data from research firm OPIS. After expenses, stations may keep roughly 15 cents per gallon, said Jeff Lenard, a vice president at NACS.
“Some make more, some make less,” Lenard said.
Patrick De Haan, head of petroleum analysis at GasBuddy, compared it to a homeowner’s role in setting their sale price.
“If I was selling a house today, I’d be beholden to whatever the housing market is,” De Haan said. “That’s the same for gas station owners. Whatever the price of oil and gasoline are, they are a price taker, not maker.”
Although the national average just passed $4 a gallon, the price that drivers pay varies widely by state, city and station.
Taxes alone can create large gaps. California's gas taxes and fees totaled about 71 cents per gallon last year, compared with roughly 9 cents in Alaska.
Distance from refineries, the type of retailer, how much volume the location goes through and whether there are other fuel options nearby also play a role.
Gas stations near competitors may choose to price gasoline competitively on large outdoor signs to attract drivers, hoping they'll come inside and buy higher-margin items, said Neal Walters, a partner focused on energy at the global management consulting firm Kearney.
“It’s one of the only retail locations where you don’t have to go into the store to find out what you’re paying,” Walters said.
While U.S. retailers sell hundreds of millions of gallons of gas a day nationwide, they typically won't see large gains when prices rise.
“The margins shrink when prices go up because it’s harder for them to pass along the increases as quickly as they themselves get them,” De Haan of GasBuddy said.
When oil prices start to fall, retailers may recover some of those losses, particularly if there’s uncertainty about future supply costs. Prices can rocket up but tend to drift down like a falling feather, said Garrett Golding, assistant vice president for energy programs at the Federal Reserve Bank of Dallas.
Higher gas prices can also hurt sales inside the gas stations, if customers who are being squeezed at the pump spend less on other things.
“So it’s not always the case that higher prices mean the service station owners are actually doing better,” Golding said.
Most profits in the oil and gas supply chain are made upstream, he said, by companies that extract and refine crude oil. Still, Golding says they aren't necessarily celebrating; at some point, a significant spike in prices could start to hurt demand.
“It may be a good stretch of days or weeks for them,” he said, “but they’re also cautious of what it could portend.”

Gas prices are displayed at a Chevron gas station, in downtown Los Angeles, Tuesday, March 31, 2026. (AP Photo/Jae C. Hong)
BOSTON--(BUSINESS WIRE)--Apr 2, 2026--
Seaport Therapeutics (“Seaport” or the “Company”), a clinical-stage therapeutics company advancing novel neuropsychiatric medicines with a proven strategy and team, today announced positive topline data from its single-ascending dose (SAD) and crossover portions of its Phase 1 proof-of-concept clinical trial evaluating GlyphAgo™ (SPT-320™ or Glyph Agomelatine), a novel, Glyphed oral prodrug of agomelatine in development for generalized anxiety disorder (GAD). The clinical proof-of-concept topline results demonstrated that GlyphAgo exceeded the program’s target of a 2-fold increase in bioavailability compared to unmodified agomelatine, achieving therapeutic levels of agomelatine at substantially lower doses that reduce liver exposure and are projected to reduce or eliminate the need for liver function testing.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260402742148/en/
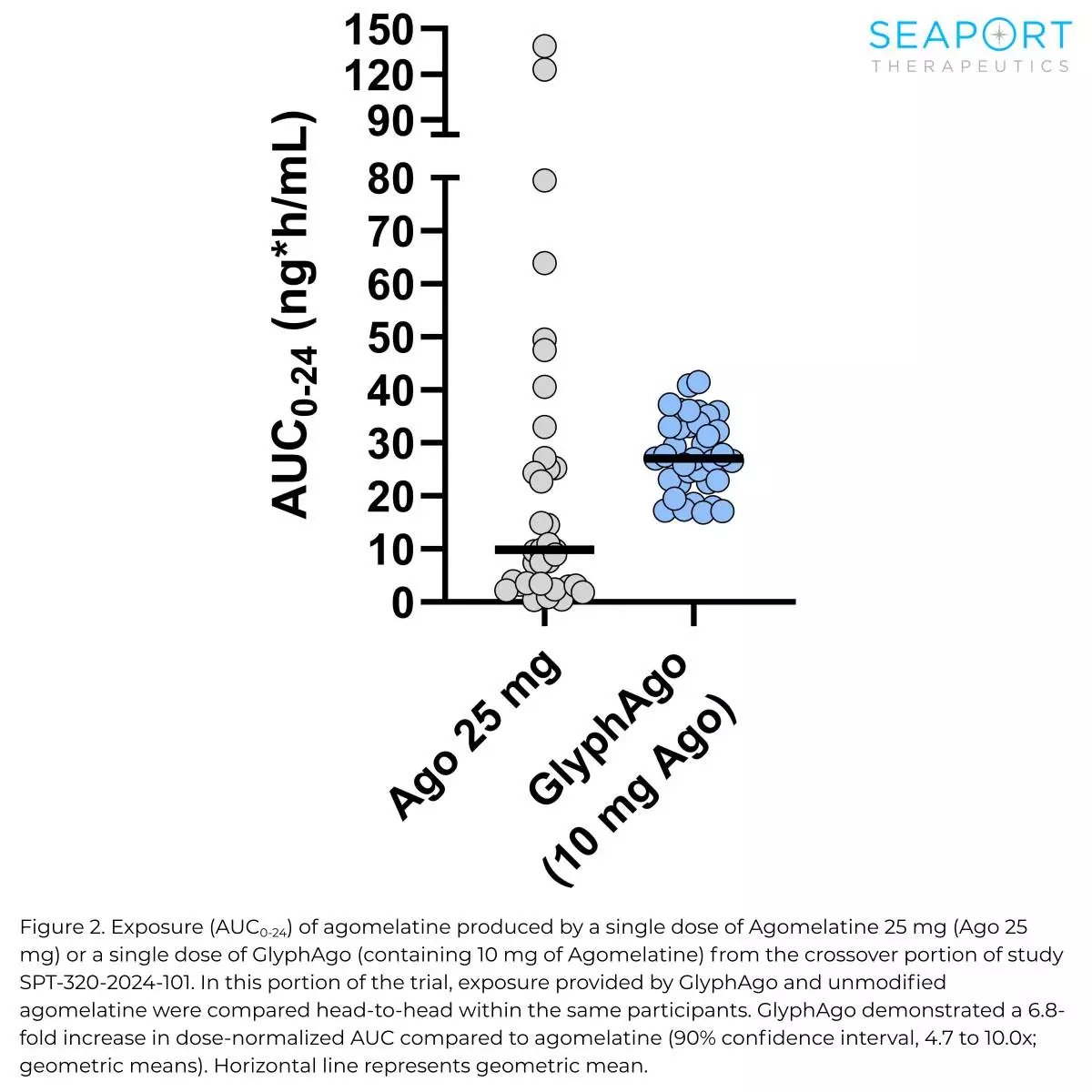
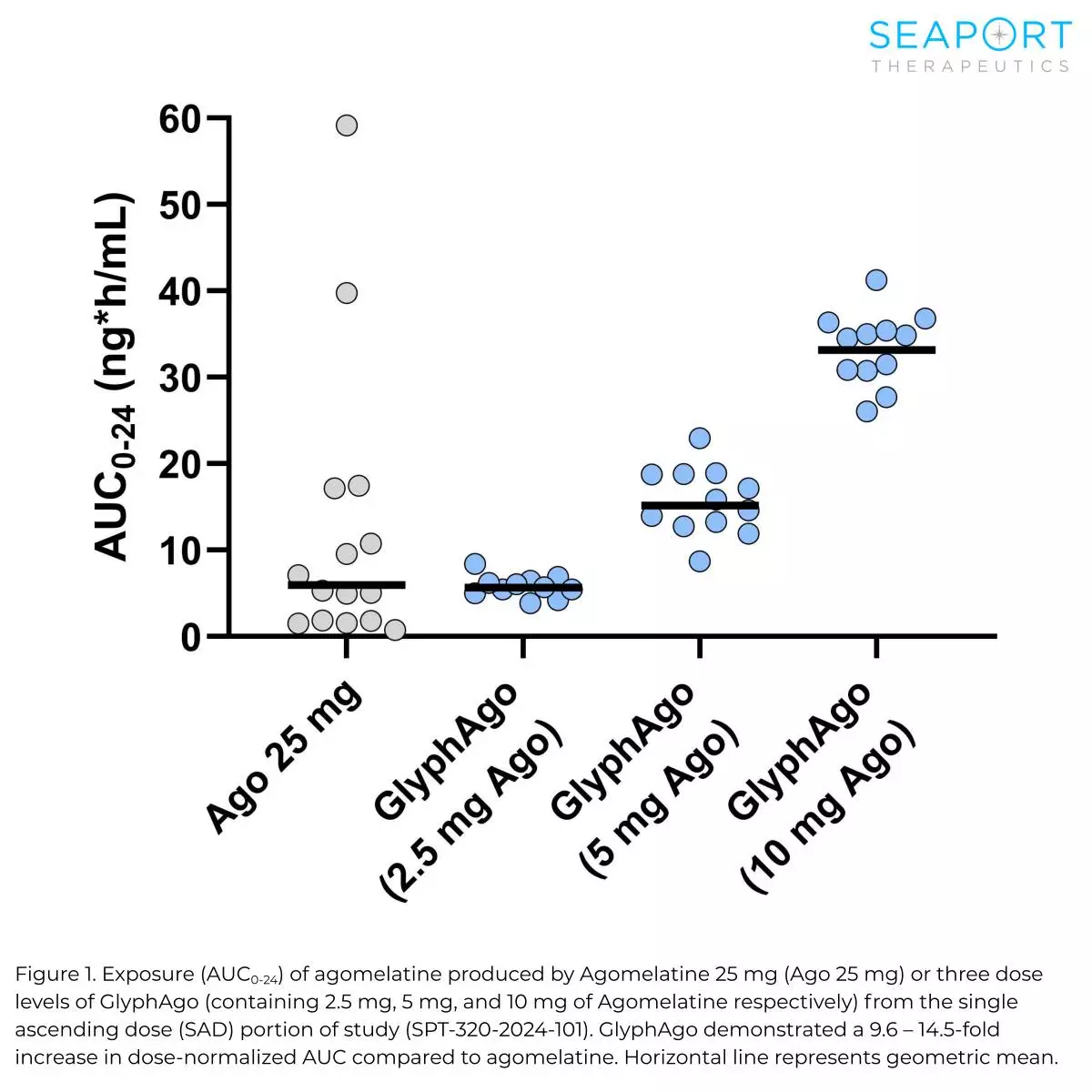
In the head-to-head crossover portion of the trial, GlyphAgo demonstrated a 6.8-fold increase in bioavailability of agomelatine compared to unmodified orally administered agomelatine. GlyphAgo also showed significantly lower (10-fold) PK variability compared to unmodified agomelatine. The crossover portion included participants who were taking estrogen-containing oral contraceptives that are known to increase agomelatine exposure due to liver drug-drug interaction. In contrast, GlyphAgo exposure was unaffected by oral contraceptives, further supporting the ability of GlyphAgo to bypass first-pass liver metabolism. GlyphAgo demonstrated a 9.6 to 14.5-fold increase in dose-normalized exposure compared to agomelatine in a separate SAD portion of the trial in which no participants were on oral contraceptives. GlyphAgo was well tolerated and no liver-related adverse events (AEs) were observed. The completed SAD and crossover portions of the trial, which included approximately 130 participants, conclude the PK proof-of-concept objectives of the trial, while the ongoing multiple-ascending dose (MAD) portion of this trial – conducted with only GlyphAgo – is intended to further characterize the safety and PK of repeat-dosing of GlyphAgo.
“These topline data, from a well-powered Phase 1 trial, strengthen our conviction in GlyphAgo’s potential and provide further clinical validation for the Glyph platform,” said Daphne Zohar, Co-Founder and Chief Executive Officer at Seaport Therapeutics. “Based on these data, we plan to advance GlyphAgo into two parallel trials, a Phase 2a proof-of-pharmacology trial to evaluate the potential sleep benefit of GlyphAgo in patients with GAD, and a Phase 2b trial in GAD, that is a randomized placebo-controlled trial designed to be registration-enabling. We believe that GlyphAgo has the potential to bring patients with generalized anxiety disorder what could be the first new therapy in decades in the U.S. for this underserved and debilitating disorder.”
Agomelatine, a clinically validated MT1/MT2 melatonin receptor agonist and serotonin 2C (5-HT2C) receptor antagonist, is an effective anxiolytic and antidepressant approved for the treatment of GAD in Australia and major depressive disorder (MDD) in Australia and the European Union (EU). Agomelatine’s label in both Australia and the EU requires liver function testing before initiating treatment, during treatment, and upon increasing the dose. Agomelatine is not approved in the U.S.
“In GAD, agomelatine has demonstrated robust and statistically significant separation from placebo in four third-party placebo-controlled studies 1-4 and has been observed in meta-analysis to have better efficacy and tolerability than selective serotonin-reuptake inhibitors or benzodiazepines,” said Steven Paul, M.D., Co-Founder and Board Chair at Seaport Therapeutics. “Despite this positive profile, over 90 percent of unmodified agomelatine is lost to first-pass metabolism and its use has been limited by dose-dependent liver enzyme elevations. The enhanced pharmaceutical properties of GlyphAgo and resulting markedly reduced inter-individual variability in systemic exposure to agomelatine support our clinical development of GlyphAgo in GAD.”
Using Seaport’s proprietary Glyph™ platform, GlyphAgo is designed to enhance lymphatic absorption and avoid first-pass liver metabolism, thereby enhancing oral bioavailability and reducing side effects. By leveraging an alternative absorption pathway via the intestinal lymphatic system used by dietary fats, GlyphAgo is designed to increase systemic exposure of agomelatine, enabling exposure levels of agomelatine that are effective in GAD but at a lower dose that reduces liver exposure and reduces or eliminates the need for liver function testing. Based on the data that Seaport has generated to date, GlyphAgo has the potential to become a leading treatment for GAD.
“Agomelatine combines a differentiated mechanism with a favorable efficacy and tolerability profile in GAD, but its potential has been previously limited by first-pass liver metabolism and the need for burdensome liver testing,” said Daniel Bonner, Ph.D., Co-Founder and Senior Vice President, Platform, at Seaport Therapeutics. “These results show that GlyphAgo exceeded the targeted improvement in bioavailability, achieving robust exposure, and a more consistent PK profile at a substantially lower dose of agomelatine.”
Phase 1 Trial Design
The Phase 1 proof-of-concept trial is being conducted in multiple parts to evaluate the safety, tolerability, and PK of GlyphAgo and to compare the PK of GlyphAgo to agomelatine alone. The trial includes single and multiple-ascending dose cohorts, as well as a crossover portion, (including both food-effect and within-participant comparison between GlyphAgo and agomelatine), using both open-label and placebo-controlled designs. In the SAD portion of the Phase 1 trial, healthy volunteers received a single administration of either ascending doses of GlyphAgo or a 25 mg dose of agomelatine, an approved efficacious dose in Australia and the EU, to assess PK parameters, including area under the curve (AUC), a measure of overall exposure, and Cmax, or peak plasma concentrations. In the SAD portion, participants were healthy volunteers with no evidence of liver impairment who were not taking any medications or supplements known to alter the PK of agomelatine, including fluvoxamine or estrogen-containing oral contraceptives. In the crossover portion, participants were randomized to one of two sequences designed to assess the food effect on a single dose level of GlyphAgo and compare it with a 25 mg dose of agomelatine. Both sequences evaluated GlyphAgo under fed and fasted conditions before agomelatine, a structure chosen to avoid confounding GlyphAgo safety results with agomelatine’s known liver toxicity.
Topline Results
Crossover Portion
SAD Portion
Seaport plans to present additional analyses from the Phase 1 trial, including the results from the MAD portion, at future upcoming scientific meetings.
About GlyphAgo (SPT-320 or Glyph Agomelatine)
GlyphAgo is a novel, Glyphed oral prodrug of agomelatine, a clinically validated anxiolytic and antidepressant that is approved for the treatment of GAD in Australia and MDD in Australia and the EU. Using Seaport’s proprietary Glyph platform, GlyphAgo is designed to avoid first-pass liver metabolism and increase systemic exposure of agomelatine, enabling exposure levels of agomelatine that are effective in GAD but at a lower dose that reduces liver exposure and reduces or eliminates the need for liver function testing. Based on internal analyses, Seaport believes a two-fold increase in the bioavailability of agomelatine with GlyphAgo dosing will reduce or eliminate liver enzyme elevations. Based on the data generated to date, Seaport believes GlyphAgo has the potential to become a leading treatment for GAD.
About Seaport Therapeutics
Seaport Therapeutics is a clinical-stage therapeutics company advancing the development of novel neuropsychiatric medicines in areas of high unmet patient needs. The Company has a proven strategy of advancing clinically validated mechanisms previously held back by limitations that are overcome with its proprietary Glyph technology platform. All the therapeutic candidates in its pipeline of first and best-in-class medicines are based on the Glyph platform, which is uniquely designed to enable oral bioavailability, bypass first-pass metabolism and reduce liver enzyme elevations or hepatotoxicity and other side effects. Seaport is led by an experienced team that invented and advanced important neuropsychiatric medicines and is guided by an extensive network of renowned scientists, clinicians, and key opinion leaders. For more information, please visit www.seaporttx.com.
Seaport Therapeutics Announces Positive Proof of Concept Topline Results from Ongoing Phase 1 Trial of GlyphAgo™ in Healthy Volunteers
Seaport Therapeutics Announces Positive Proof of Concept Topline Results from Ongoing Phase 1 Trial of GlyphAgo™ in Healthy Volunteers












