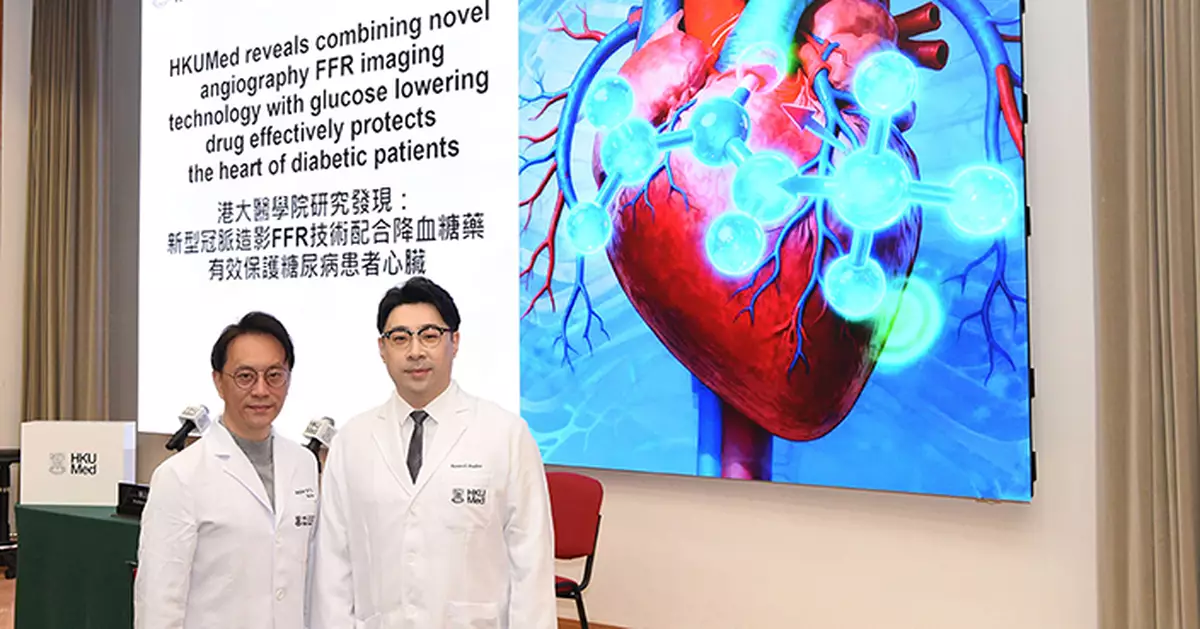
A recent study led by the Department of Medicine, School of Clinical Medicine, LKS Faculty of Medicine of the University of Hong Kong (HKUMed), in collaboration with the University of Hong Kong-Shenzhen Hospital, has demonstrated that combining an innovative coronary imaging technology known as the caFFR system, with diabetes drug SGLT2 inhibitors can significantly reduce the risks of major adverse cardiovascular events (MACE), heart failure and death among patients with type 2 diabetes mellitus (T2DM) and coronary artery disease. This dual-pronged strategy provides a precise measurement of coronary blood flow while lowering glucose levels, offering an effective approach to treating exceptionally high-risk patients. The findings were published in the Diabetes and Metabolism Journal link to the publication.
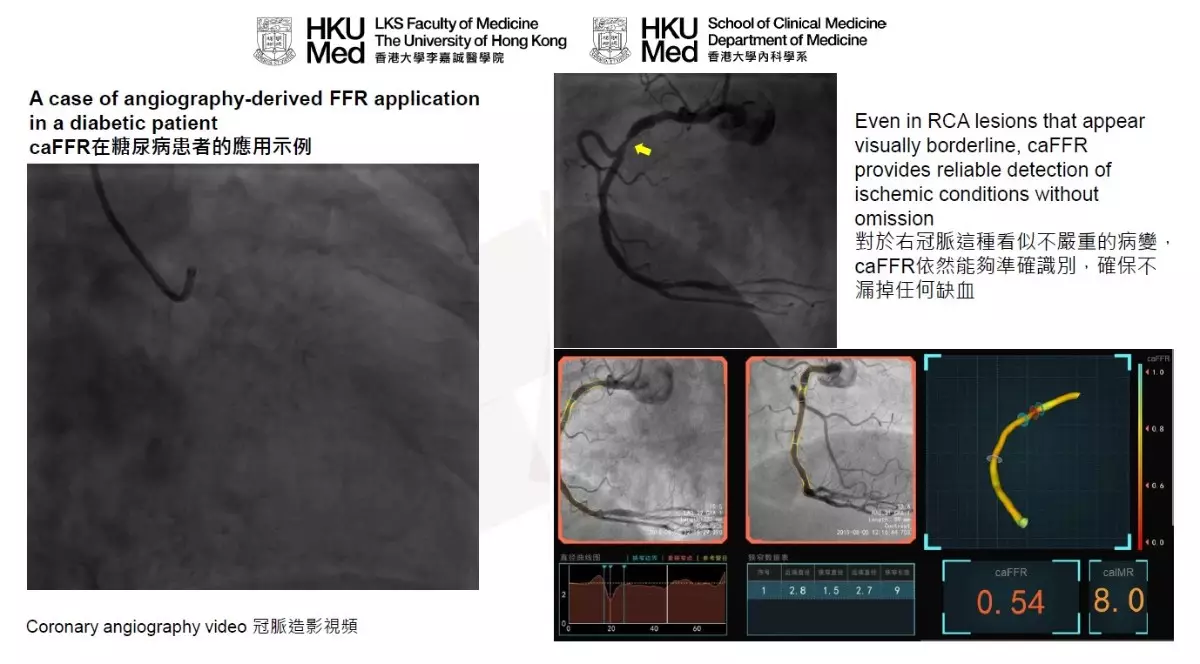
caFFR images access coronary blood flow
Patients with T2DM commonly develop more complex and severe forms of coronary artery disease, often involving multiple narrowed or blocked arteries. This complexity makes it challenging for cardiologists to achieve 'complete revascularisation', where all significantly blocked arteries are fully opened to restore blood flow. Without detailed functional assessment, some blockages that appear mild on imaging but are functionally important may be overlooked. If these high-risk blockages are not identified, patients may be at risk of 'incomplete revascularisation' and experience persistent ischaemia despite treatment, ultimately increasing their long-term cardiac risk.
To address this challenge, Professor Yiu Kai-hang, Clinical Professor in the Department of Medicine, School of Clinical Medicine, HKUMed, led a research team to evaluate the use of the caFFR system, an innovative imaging technology that allows accurate measurement of coronary blood flow from standard angiogram images. This technique enables cardiologists to identify which arterial blockages are truly responsible for ischaemia, thus supporting more precise decision-making and the development of more effective interventional treatment strategies for high-risk patients.

Professor Yiu Kai-hang explains that the functional assessment using the caFFR system is crucial for achieving optimal revascularisation in diabetic patients. SGLT2 inhibitors offer robust cardiovascular protection and significantly improve survival outcomes, even in cases of incomplete revascularisation.
SGLT2 inhibitors provide powerful cardiac protection
The study analysed data from 671 patients with both T2DM and coronary artery disease who underwent angiogram imaging in public hospitals between 2014 and 2016. While complete revascularisation was achieved in some patients, many still had residual stenosis after undergoing the revascularisation procedure due to diffuse and complex diabetic atherosclerosis. Remarkably, for those with incomplete revascularisation, the use of SGLT2 inhibitors provided powerful vascular protecton. The three-year incidence of MACE was markedly reduced from 17.8% to 8.3%, while all-cause mortality dropped sharply from 16.3% to 6.3% over the same period.

HKUMed reveals that combining novel caFFR imaging technology with glucose lowering drug effectively protects the heart of diabetic patients. In the photo are Professor Yiu Kai-hang (left), who led the research, and his team member Dr Xuan Haochen.
'Our findings show that functional assessment using the caFFR system allows clinicians to accurately identify blockages that truly cause ischemia, which is crucial for achieving optimal revascularisation in diabetic patients,' said Professor Yiu Kai-hang. 'Moreover, even when complete revascularisation cannot be achieved, SGLT2 inhibitors offer robust cardiovascular protection, significantly improving survival outcomes. This dual approach represents a major step forward in managing heart disease among individuals with diabetes.'
The study underscores the complementary roles of precision interventional imaging and pharmacological therapy in improving outcomes for diabetic patients with coronary artery disease. By integrating caFFR-guided vascular reconstruction procedures with SGLT2 inhibitor therapy, clinicians can better tailor treatment to each patient's needs, offering both anatomical and metabolic protection for the heart.
Professor Yiu added, 'This study has importance implications for clinical practice. It demonstrates that even when structural risks in the blood vessels may persist, SGLT2 inhibitors provide a vital safety net, reducing future cardiovascular risks. The findings reinforce HKUMed's ongoing commitment to translating clinical innovation into better patient care.'
About the research team
The study was led by Professor Yiu Kai-hang, Clinical Professor, Department of Medicine, School of Clinical Medicine, HKUMed, and conducted in collaboration with the University of Hong Kong–Shenzhen Hospital.