BOSTON--(BUSINESS WIRE)--Apr 30, 2026--
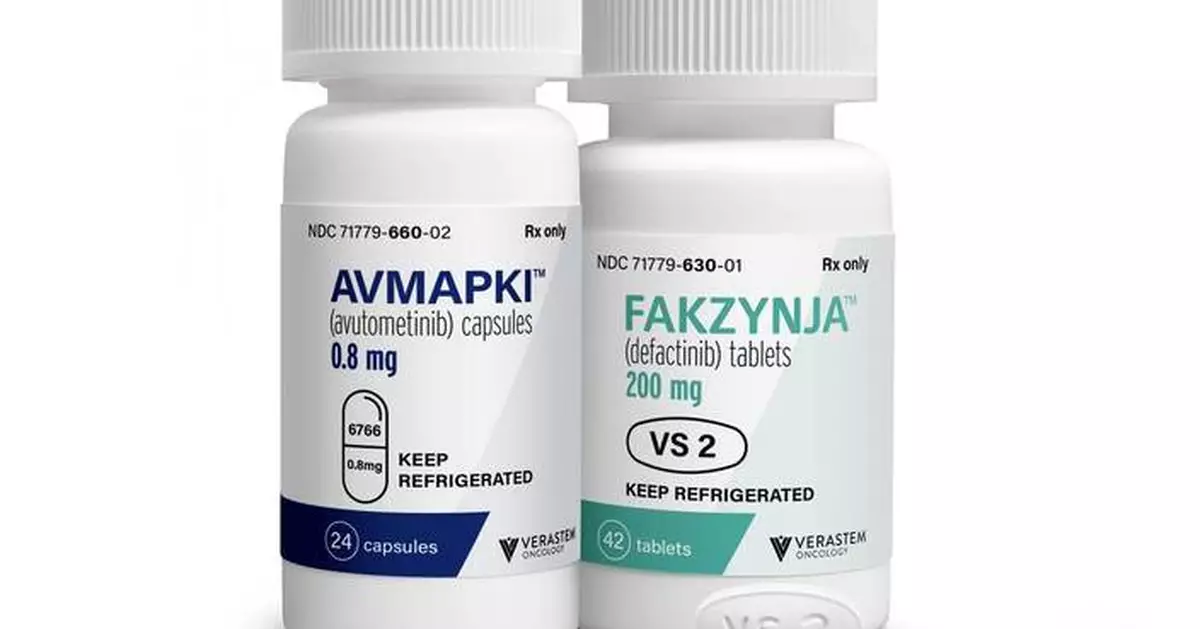
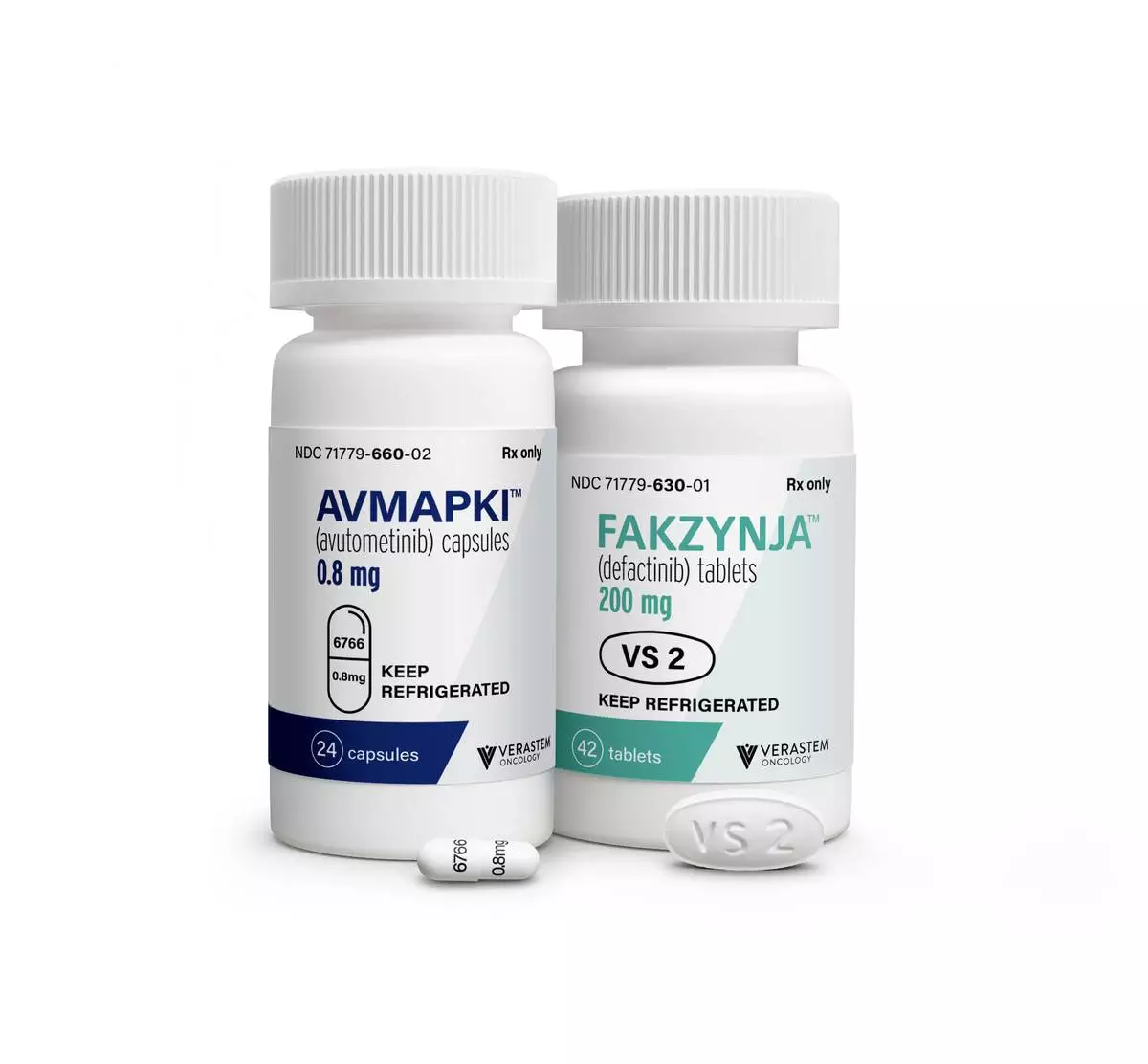
Verastem Oncology (Nasdaq: VSTM), a biopharmaceutical company committed to advancing new medicines for patients with RAS/MAPK pathway-driven cancers, today announced the launch of its Reimagine Recurrent Low-Grade Serous Ovarian Cancer (LGSOC) campaign for AVMAPKI ® FAKZYNJA ® CO-PACK (avutometinib capsules; defactinib tablets), the first and only treatment specifically approved by the U.S. Food and Drug Administration (FDA) for adult patients with KRAS-mutated recurrent LGSOC who have received prior systemic therapy. The integrated campaign encourages healthcare professionals (HCPs) and patients to rethink what may be possible at and beyond the earliest recurrence for people living with KRAS-mutated recurrent LGSOC.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20260428101589/en/
LGSOC is a rare and distinct form of ovarian cancer, with unique biology and clinical behavior different from other ovarian cancers. Its tumor growth is primarily driven by the RAS/MAPK pathway with activating mutations, most commonly in the KRAS gene, found in many cancers that often correlate with poor prognosis. Approximately 30% of patients with LGSOC have a KRAS mutation, making it the most common type of gene mutation in the disease. Patients with LGSOC often experience delays in diagnosis and have historically had limited treatment options specifically for their disease.
“The Reimagine Recurrent LGSOC campaign shifts how we think about KRAS-mutated recurrent LGSOC treatment and supporting both patients and healthcare professionals in rethinking what may be possible at and beyond the earliest recurrence,” said Dan Paterson, president and chief executive officer at Verastem Oncology. “The FDA’s accelerated approval of AVMAPKI FAKZYNJA CO-PACK marked an important step forward for patients with KRAS-mutated recurrent LGSOC, underscoring the need for treatments specifically developed for this population. With this campaign, we’re building on that momentum to further inform treatment decisions and support patients throughout their care journey.”
The Reimagine campaign was informed by insights from patients and HCPs, highlighting the need for both emotional resonance and practical, accessible information. For HCPs, the campaign encourages re-evaluation of treatment approaches in the context of a treatment specifically approved by the FDA for KRAS-mutated recurrent LGSOC. For patients, it reflects a desire not only to manage their condition, but to envision a future shaped by progress and meaningful life milestones.
“Patients with KRAS-mutated recurrent LGSOC have historically had limited resources tailored to their specific experience,” said Linda Stamler, vice president of marketing at Verastem Oncology. “The Reimagine Recurrent LGSOC campaign was developed to help identify and support patients who may be appropriate for treatment with AVMAPKI FAKZYNJA CO-PACK. Through this campaign, we are offering resources such as patient stories, tools to help manage side effects, videos explaining mechanism of action and dosing, and guides to help set treatment expectations so patients and HCPs feel more informed and supported.”
For more information, HCPs can visit https://www.avmapkifakzynjaco-pack-hcp.com/ and patients can visit https://www.avmapkifakzynjaco-pack.com/.
About Low-Grade Serous Ovarian Cancer (LGSOC)
LGSOC is a rare ovarian cancer that is insidious and persistent. LGSOC is distinct and different from high-grade serous ovarian cancer (HGSOC) and requires different treatment. LGSOC is highly recurrent and less sensitive to chemotherapy compared to HGSOC. Approximately 6,000-8,000 women in the U.S. and 80,000 worldwide are living with this disease. LGSOC affects younger women with bimodal peaks of diagnosis at ages between 20-30 and 50-60 and has a median survival of approximately ten years. Approximately 70 percent of LGSOC shows RAS pathway-associated mutations, and 30 percent of people with LGSOC have a KRAS mutation. The majority of patients report a negative impact of LGSOC on their mental and physical health, fertility, and long-term quality of life.
About AVMAPKI and FAKZYNJA Combination Therapy
AVMAPKI (avutometinib) inhibits MEK kinase activity while also blocking the compensatory reactivation of MEK by upstream RAF. RAF and MEK proteins are regulators of the RAS/RAF/MEK/ERK (MAPK) pathway. Blocking RAF and/or MEK activates FAK, a key mediator of drug resistance. FAKZYNJA (defactinib) is a FAK inhibitor and together, the avutometinib and defactinib combination was designed to provide a more complete blockade of the signaling that drives the growth and drug resistance of RAS/MAPK pathway-dependent tumors.
The U.S. Food and Drug Administration (FDA) approved AVMAPKI ® FAKZYNJA ® CO-PACK (avutometinib capsules; defactinib tablets) for the treatment of adult patients with KRAS-mutated recurrent LGSOC who have received prior systemic therapy on May 8, 2025. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. Verastem is conducting RAMP 301 (GOG-3097/ENGOT-ov81/GTG-UK) (NCT06072781), an international Phase 3 confirmatory trial evaluating the combination of avutometinib and defactinib versus standard chemotherapy or hormonal therapy for the treatment of recurrent low-grade serous ovarian cancer (LGSOC) with and without a KRAS mutation. Verastem is also evaluating avutometinib plus defactinib with standard-of-care chemotherapy as a potential treatment in the first-line for patients with advanced pancreatic cancer (RAMP 205; NCT05669482). Avutometinib and defactinib are not approved by the FDA or any other regulatory authority, either in combination or with other therapies, for any of these investigative uses. Neither avutometinib nor defactinib are approved by the FDA or any other regulatory authority on a stand-alone basis for any use.
AVMAPKI FAKZYNJA CO-PACK U.S. Indication
Indication
AVMAPKI FAKZYNJA CO-PACK is indicated for the treatment of adult patients with KRAS -mutated recurrent low-grade serous ovarian cancer (LGSOC) who have received prior systemic therapy.
This indication is approved under accelerated approval based on tumor response rate and duration of response . Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
Important Safety Information
Warnings and Precautions
Adverse Reactions
The most common (≥ 25%) adverse reactions, including laboratory abnormalities, were increased creatine phosphokinase, nausea, fatigue, increased aspartate aminotransferase, rash, diarrhea, musculoskeletal pain, edema, decreased hemoglobin, increased alanine aminotransferase, vomiting, increased blood bilirubin, increased triglycerides, decreased lymphocyte count, abdominal pain, dyspepsia, dermatitis acneiform, vitreoretinal disorders, increased alkaline phosphatase, stomatitis, pruritus, visual impairment, decreased platelet count, constipation, dry skin, dyspnea, cough, urinary tract infection, and decreased neutrophil count.
Drug Interactions
Use in Specific Populations
Click here for full Prescribing Information.
About Verastem Oncology
Verastem Oncology (Nasdaq: VSTM) is a biopharmaceutical company committed to developing and commercializing new medicines to improve the lives of patients diagnosed with RAS/MAPK pathway-driven cancers. Verastem markets AVMAPKI ® FAKZYNJA ® CO-PACK in the U.S. Our pipeline is focused on novel small molecule drugs that inhibit critical signaling pathways in cancer that promote cancer cell survival and tumor growth, including RAF/MEK inhibition, FAK inhibition, and KRAS G12D inhibition. For more information, please visit www.verastem.com and follow us on LinkedIn.
Forward-Looking Statements
This press release includes forward-looking statements. These forward-looking statements generally can be identified by the use of words such as “anticipate,” “expect,” “plan,” “could,” “may,” “believe,” “estimate,” “forecast,” “goal,” “project,” and other words of similar meaning. Such forward-looking statements address various matters about, among other things, Verastem Oncology’s programs and product candidates, strategy, future plans and prospects, including statements related to the potential for and timing of commercialization of product candidates, the expected outcome and benefits of the Company’s collaboration with GenFleet Therapeutics (Shanghai), Inc., the timing of commencing and completing trials and compiling data, the expected timing of the presentation of data by the Company and the potential clinical value of various of the Company’s clinical trials. Each forward-looking statement contained in this press release is subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statement. Applicable risks and uncertainties include, among others: the uncertainties inherent in research and development, such as the possibility of negative or unexpected results of clinical trials; that we may not see a return on investment on the payments we have and may continue to make pursuant to the collaboration and option agreement with GenFleet, or that GenFleet may fail to fully perform under the agreement; that we may not be successful in our continued commercialization of AVMAPKI FAKZYNJA CO-PACK; that the development and commercialization of our product candidates may take longer or cost more than planned, including as a result of conducting additional studies or our decisions regarding execution of such commercialization; that data may not be available when expected; risks associated with preliminary and interim data, which may not be representative of more mature data; risks associated with the recent changes in administration policy or actions that may create regulatory uncertainty that may adversely affect our business; risks associated with the current administration’s reductions to the FDA’s workforce and any subsequent reductions that may lead to disruptions and delays in the FDA’s review and oversight of our product candidates and impact the FDA’s ability to provide timely feedback on our development programs; that our product candidates may not receive regulatory approval, become commercially successful products, or result in new treatment options being offered to patients; and the risks identified under the heading "Risk Factors" as detailed in the Company’s Annual Report on Form 10-K for the year ended December 31, 2025, as filed with the Securities and Exchange Commission (SEC) on March 4, 2026, as well as the other information we file with the SEC, are possibly realized. We caution investors not to place considerable reliance on the forward-looking statements contained in this press release. You are encouraged to read our filings with the SEC, available at www.sec.gov, for a discussion of these and other risks and uncertainties. The forward-looking statements in this press release speak only as of the date of this press release, and we undertake no obligation to update or revise any of these statements. Our business is subject to substantial risks and uncertainties, including those referenced above. Investors, potential investors, and others should give careful consideration to these risks and uncertainties.

Verastem Oncology Launches HCP and Patient Reimagine Campaign to Increase Awareness of AVMAPKI® FAKZYNJA® CO-PACK for KRAS-Mutated Recurrent Low-Grade Serous Ovarian Cancer
Verastem Oncology Launches HCP and Patient Reimagine Campaign to Increase Awareness of AVMAPKI® FAKZYNJA® CO-PACK for KRAS-Mutated Recurrent Low-Grade Serous Ovarian Cancer